The history of Arsenic’s in medicine or science has been overshadowed by its notoriety as a poison in homicides and nowadays it is being synonymous with toxicity. Arsenic species can be present in all types of environment and can originate from natural as well as anthropogenic sources. Natural sources of arsenic are: rocks with incorporated arsenic compounds, activity of volcanoes and some biological processes. Anthropogenic sources are numerous, from mining to different types of production (pesticides, wood preservatives, and pigments). Depending on oxidation state and presence in the environment, arsenic species exhibit different toxicity. Generally, arsenic (As) exists as both arsenite (AsIII) and arsenate (AsV) in natural waters or groundwater and the As(V) to As(III) ratio has been found to be in the range of 10−100 depending on the chemical nature such as pH value of water. Dangerous arsenic concentrations in ground or natural waters are considered as a 20th–21st century calamity worldwide. High arsenic concentrations in ground water are adversely affecting over 150 million people globally in the different countries like Bangladesh, India, Pakistan, China, Vietnam, Nepal, Cambodia, Myanmar, Laos, Indonesia, USA, Argentina, Chile, Mexico, Canada, Hungary, South Africa, Poland, etc. Many water sources in the world containing high concentration of arsenic cause health problems or diseases such as cancer. Chronic long-term arsenic exposure can lead to skin effects (pigmentation, keratoses, lesions, and cancer); peripheral vascular disease; hypertension and cardiovascular diseases; cancers of the bladder, kidney, and lung; diabetes mellitus; and possible neurological effects. The World Health Organization (WHO) restrict the Maximum Contaminant Level (MCL) of arsenic in drinking water at 10 parts per billion (ppb), yet the arsenic concentration levels in many locations worldwide exceed 50 ppb.
“By harnessing functionalized gold nanoparticles, this innovation enables simple, rapid, and sensitive detection of toxic arsenic in water, offering a low-cost solution for real-time monitoring and public health protection“
Arsenic contaminated water is colorless, odorless, and tasteless, and the adverse health effects of arsenic poisoning may not become apparent until long after initial consumption. As a result, an estimated 150 million people especially villagers globally are at risk of long-term arsenic poisoning from the water they drink. The available arsenic detection technologies are inadequate due to have disadvantages like expensive, time consuming, instrument based method, laboratory set-up used, required tarried technicians, requiring off-site analysis of samples, etc. On-site real time monitoring demands the effective sensing method capable to shown response with low concentrations of arsenic in aqueous media. The researchers are intended to develop the simple procedures like colorimetric technique with advantages like availability of colorimetric kits in simple form, low-cost, easy handling, using by anyone, and especially necked eye visual detection. Noble metal nanostructures are attracting interest due to their special characteristics, including major optical field increases that generate high light scattering and absorption. Of these, gold nanoparticles have attracted much attention as a unique and advantageous optical platform due to having intense Plasmon resonance absorption (SPR) band in visible region including large surface area, stability. Moreover, the strong surface SPR of gold nanoparticles in the visible region is exquisitely sensitive to their aggregation states influenced by the ions to be detected. However, choosing the right functional groups and successful functionalization on the gold nanoparticles are important in order to impart the selective detection of arsenic in water.
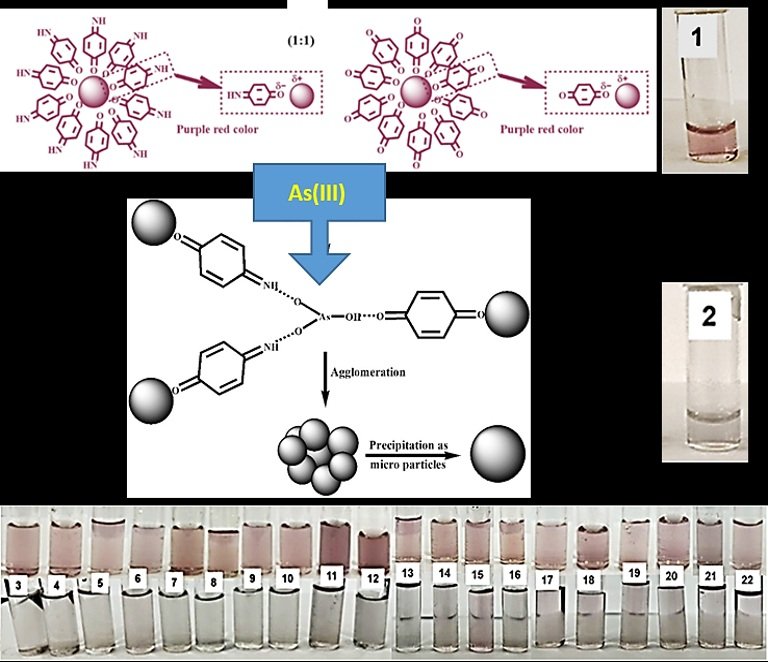
Recently, a simple, novel, rapid, selective, sensitive functionalized gold nanoparticles (AuNPs) based colorimetric method has been demonstrated for the successful sensing of arsenic ions in water. The aqueous colloid of AuNPs was synthesized separately using p-aminophenol and p-hydroquinone as reducing agents as well as stabilizers in aqueous basic medium. Au+3 ion (in Tetrachloroauric acid) was reduced to Au(0) due to oxidation of p-aminophenol to p-quinonimine (p-QI) and or p-hydroquinone to p-quinone (p-Q), respectively (see Figure). The synthesized Au(0) nanoparticles were instantly stabilized due to functionalization on the surface through electrostatic dipolar interactions with =NH functionalities of p-QI and =O functionalities of p-Q. Both the synthesized colloids of AuNPs shows a visible red-purple appearance due to SPR absorption maxima in UV-VIS spectrum at ~540 nm. The individual colloid of p-QI and p-Q functionalized AuNPs was not found to have any repose even in UV-VIS spectrum measurement towards the arsenic ion. Interestingly, the colorimetric sensing of arsenic ion was recorded in UV-VIS spectrum of equal volume (1:1) colloidal mixture of p-QI and p-Q functionalized AuNPs. Though the UV-VIS sensing response of equal volume colloidal mixture towards As(III) ions was obtained within the pH range 2-12 but the best result was observed at pH 7-8. A very similar sensing response was also recorded at pH 7-8 towards As(V) chemically reduced to As(III) using KI/SnCl2 in HCl medium. A linearity range up to 0.035 mM with sensitivity of ~16.5 mM-1 and a limit of detection of ~2.5 μM was observed towards the detection of As(III) ions. The visual detection of sensing performances towards the arsenic ions and inference effect common anions, cations, and biomolecules was tested using AuNPs colloidal mixture diluted to 1:10 (v/v). A purple-red was visualized towards the addition of common ions such as F–, Cl–, Br–, I–, HCO3-, SO42-, PO43-, CO32-, NO3-, NO2-, Ca2+, Mg2+, K+, Fe2+, Fe3+, and biomolecules such as Fructose, Sucrose, Lactose, Uric Acid, Ascorbic Acid, Dopamine (3-22 in Figure below). Whereas, an immediate decolorization of mixture of functionalized AuNPs colloid was observed from purple-red by simple addition of As(III) ions in the colloid mixture in presence of the other ions (see Figure). The dipolar interaction between the p-QI and p-Q functional groups and the AuNPs was supposed to break-up upon the addition of As(III) ions, which triggered the AuNPs to aggregate (see Figure). The visual limit of detection for the As(III) ions was evaluated as ~44 μM. It should be noted that the limit of detection was determined higher than that of the WHO recommended limit (0.133 × 10-3 mM). However, a high volume of water sample after concentrating by evaporation should be used to extend the limit of detection below the WHO recommended limit.
Therefore, on-site sensor for ions operating in 100% aqueous medium are still rarer and they suffer from poor efficiency, response, selectivity and add restrictions to their practical applications. In addition, the special emphasis should be given for the color changes, as signaling an event detected by the naked eye owing to the low cost or to avoid use of detection equipment. In order to address the above limitations, the researchers are designing metal nanoparticle based rapid as well as portable on-site optical sensor for ions in aqueous medium. To impart the ion selectivity and ultra-sensitivity, influence the strong adsorption, improve the stability, different functionalization should be introduced on the nanoparticles from nearly molecular-level. The de-stability of the nanomaterials should also be equally considered for successful sensing of any analyte through suitable strong interaction with the functional groups on the nanomaterials. Therefore, the attention should be given towards the finding of suitable functional groups on the nanomaterials surface or suitable combination of them for sensing applications.