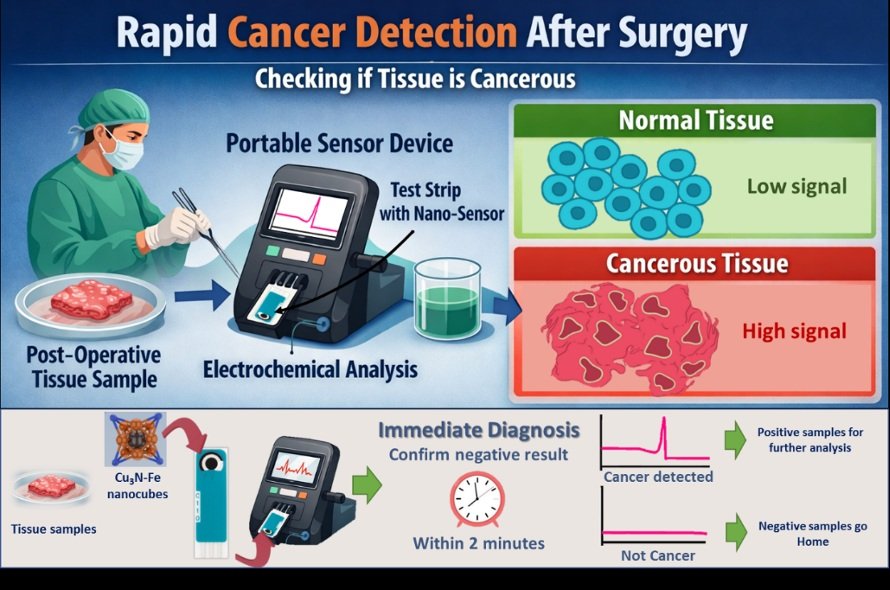
Research in our laboratory focuses on the design and application of advanced nanomaterials for rapid biosensing, disease diagnostics, and catalytic technologies. We develop various advanced nanostructures to create sensitive sensing platforms capable of detecting biologically relevant molecules. One of our recent studies demonstrated that iron-doped Cu₃N nanocubes can act as highly efficient electrochemical sensors for hydrogen peroxide, an important metabolic marker released by cancer cells. By integrating these nanomaterials with simple electrode systems, we aim to create rapid diagnostic tools that can distinguish cancerous tissues from non-cancerous samples within minutes. Beyond cancer diagnostics, our laboratory also explores nanotechnology for biosensing of pathogens, targeted drug delivery, and catalytic applications, with the broader goal of developing affordable and point-of-care technologies that can make advanced healthcare and diagnostics more accessible.
“What if we could reveal within minutes whether a piece of tissue is cancerous, simply by detecting subtle chemical signals released by cells? Our research in nanotechnology suggests that this idea is quickly becoming a reality.“
Cancer remains one of the most challenging diseases of our time, not only because of its biological complexity but also because of the difficulty of detecting it early and accurately. While modern molecular diagnostics have significantly improved our ability to identify cancer, many of these techniques such as polymerase chain reaction (PCR), advanced imaging, or sequencing require expensive instrumentation, trained personnel, and centralized laboratory facilities. As a result, there is a growing global demand for diagnostic technologies that are rapid, affordable, and capable of functioning at the point of care. In my view, nanomaterial-based biosensors represent one of the most promising paths toward achieving this goal. Our recent work focuses on developing a rapid electrochemical platform that can detect metabolic biomarkers associated with cancer cells within minutes. One of the key metabolic signatures of cancer progression is the abnormal production of reactive oxygen species (ROS), particularly hydrogen peroxide (H₂O₂). Cancer cells often exhibit elevated oxidative stress and altered metabolic pathways, leading to higher levels of extracellular hydrogen peroxide compared to normal cells. This biochemical difference provides a unique opportunity: if we can measure H₂O₂ quickly and selectively, it may serve as a simple indicator of cancer-related metabolic activity. To address this challenge, our research team developed a nanomaterial-based sensing platform using iron-doped copper nitride (Cu₃N-Fe) nanocubes. The nanocomposite is drop-cast onto a disposable carbon electrode, creating an electrochemically active surface capable of rapidly oxidizing hydrogen peroxide.
To explore its potential real-world relevance, we evaluated the platform using clinical tissue samples. The results showed that the sensor could successfully differentiate cancerous tissues from adjacent non-cancerous tissues with a high degree of accuracy. While these findings are still part of an early-stage investigation, they highlight the potential of nanomaterial-based electrochemical platforms as rapid diagnostic tools for cancer screening and biopsy analysis. Beyond the specific system we developed, I believe this work reflects a broader transformation occurring in the field of biomedical diagnostics. Traditionally, diagnostic technologies have focused on identifying genetic mutations or protein markers associated with disease. While these approaches remain essential, metabolic biomarkers offer an additional and often faster route to detection. Cellular metabolism responds rapidly to disease states, and these biochemical changes can often be measured using relatively simple analytical tools. By combining metabolic biomarkers with advanced nanomaterials, we can create highly sensitive sensors that operate in real time. Nanomaterials are uniquely suited for this purpose. Their extremely high surface-to-volume ratios, tunable electronic properties, and ability to be engineered at the atomic scale allow researchers to design materials with tailored catalytic and sensing capabilities. Over the past decade, advances in nanotechnology have enabled the development of a wide variety of sensing platforms based on metal nanoparticles, carbon nanostructures, semiconductor nanocrystals, and hybrid nanocomposites. These systems can detect a wide range of biological targets, including nucleic acids, proteins, metabolites, and pathogens. However, the true impact of these technologies will depend on their ability to move beyond laboratory demonstrations and into practical clinical applications. In many parts of the world, especially in resource-limited settings, access to advanced diagnostic facilities remains limited. Point-of-care diagnostic devices that are portable, inexpensive, and easy to use could dramatically improve early detection and treatment outcomes. Electrochemical biosensors offer several advantages for such applications: they are low-cost, require minimal sample preparation, and can be integrated into compact electronic devices. Smartphone-based readout systems, miniaturized potentiostats, and wireless data transmission could allow diagnostic results to be obtained and shared instantly. Such systems could be especially valuable in rural healthcare environments where centralized laboratories are not readily accessible. Artificial intelligence and data-driven approaches will also play an increasingly important role. As sensing technologies generate larger and more complex datasets, machine learning algorithms can help identify patterns, improve diagnostic accuracy, and predict disease progression. The integration of nanotechnology, biosensing, and computational analysis may ultimately lead to highly personalized diagnostic systems tailored to individual patients.
For young researchers entering this field, I believe the most important lesson is the value of interdisciplinary thinking. Modern biomedical challenges rarely fall within a single discipline. Progress often emerges at the intersection of chemistry, physics, biology, materials science, and engineering. In our own work, advances in nanomaterial synthesis, electrochemistry, and cellular biology all contributed to the development of the sensing platform. Cultivating the ability to communicate and collaborate across these fields is essential. tform. Cultivating the ability to communicate and collaborate across these fields is essential. Finally, researchers should remember that the ultimate goal of biomedical innovation is to improve human health. Technologies that are elegant in the laboratory must also be practical in real-world settings. Designing affordable, scalable, and accessible diagnostic solutions will be critical for addressing global health challenges in the decades ahead. Nanomaterial-based biosensors hold tremendous promise for transforming the landscape of medical diagnostics. By exploring their properties of engineered nanomaterials and combining them with sensitive detection strategies, we can begin to develop rapid, point-of-care tools capable of identifying disease-related biomarkers within minutes. While significant work remains before such technologies become widely available in clinical practice, the progress achieved so far demonstrates that the future of diagnostics may lie in small, intelligent materials capable of delivering powerful insights into the biology of disease. The coming decade will likely witness rapid advances in nanotechnology, biosensing, and digital healthcare. If researchers continue to collaborate across disciplines and focus on translating scientific discoveries into practical solutions, we may soon see a new generation of diagnostic tools that bring fast, affordable cancer detection directly to the patient’s bedside.