What are aqueous zinc-ion batteries, and why are scientists interested in them today?
In response to global climate change, rechargeable batteries have a pivotal role in industrial decarbonization and the energy transition to a sustainable energy storage landscape. Owing to the high demand of electric vehicles and consumer electronics, in the last couple of decades lithium-ion batteries (LIB) have exhibited impressive performance and thus captured the whole market; however, scarcity, complicated extraction processes, and safety issues call for new types of rechargeable batteries that allow cleaner, safer, and more efficient energy storage solutions. Aqueous zinc-ion batteries (AZIBs) offer a rechargeable and sustainable energy storage system with good capacity (gravimetric: 820 mA h g-1 and volumetric: 5854 mA h cm-3), low production cost, and high safety. It could provide an ideal technology for stationary and large-scale such as grid-scale energy storage. Additionally, AZIBs can be used in flexible batteries for wearable electronics and biomedical applications. AZIBs comprise Zn metal as an anode, which is abundant, low cost, and safe to handle, and transition metal oxides such as manganese or vanadium oxide, Prussian blue analogues, and organic compounds as a cathode. Unlike LIBs, where toxic and flammable organic solvents are used as electrolytes, AZIBs use water-based electrolytes, preventing fire hazards common in LIBs. Because of their safety, low cost, and sustainability, AZIBs are gaining strong interest as next-generation energy storage systems.
What are the main problems that currently limit the performance or lifetime of zinc batteries?
Aqueous zinc ion batteries (AZIBs) face critical challenges primarily driven by water-based electrolytes, including limited operating voltage due to low water decomposition voltage (1.23 V), severe zinc dendrite formation, hydrogen evolution reaction (HER), surface passivation (corrosion), and cathode dissolution. These issues cause short circuits, low coulombic efficiency, poor cycling stability, and limited energy density. On the anode side, non-uniform deposition of Zn, known as Zn-dendrites, occurs during charging, while at the anode-electrolyte interface, HER leads to the corrosion of Zn metal during charging, discharging, and rest periods. In the cathode, as expected, zinc ions (Zn2+) would act as primary charge carriers during (de)intercalation in the host materials; however, protons (H+) are found to be co-carriers in some cases.
Even proton storage may become predominant at high rates and lower potentials. Proton co-insertions create problems for prolonged cycling as it produces hydroxyl (OH–) anions via water decomposition, which increases the pH to facilitate the formation of unwanted layered zinc hydroxide sulphate (ZSH) depositing on the cathode (anode) surfaces. Structural instability and capacity fading of the cathode, particularly metal (Mn and V) oxides are due to irreversible reactions leading to phase change and dissolution of redox-active materials in electrolytes during stripping/plating.
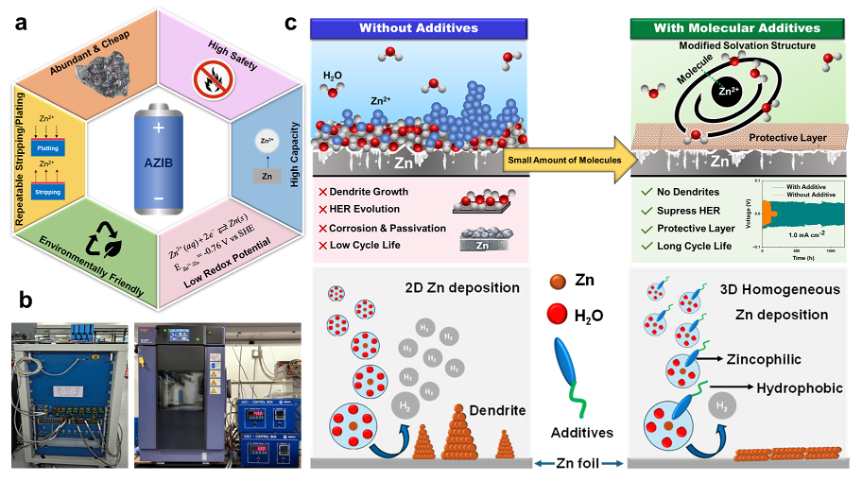
Your research talks about “molecular materials.” In simple words, what are these molecules and how do they work inside a battery?
Molecular materials include small molecules or their self-assembled structures, gels, polymers, supramolecular architecture, hydrogen-bonded organic frameworks (HOFs), covalent organic frameworks (COFs), metal organic frameworks (MOFs), ionic liquids, organic–inorganic hybrid materials, biomolecules such as vitamins, proteins, polysaccharides, inorganic molecules, etc. which are generally built from functional molecules via a “bottom–up” approach that retains their properties down to the molecular scale.
Enhancement of Zn/Zn2+ reversibility largely relies on the cooperative functions of all components of the battery, such as cathode, anode, electrolytes, and separators. Any hindrance in the synchronizations of the functional components leads to shortening of the life cycle and capacity fading of the battery. Molecular materials have multitudes of functionalities that can enhance the lifetime of the battery in several ways, such as modifying electrode surface, solvation structure, desolvation kinetics, and ionic conductivity, and alleviating the side reactions and HER at the electrode and electrolyte interface.
For example, a small organic molecule, 4-methyl pyridine N-oxide, having a zincophilic bipolar head and hydrophobic tail can make a Zn complex with a polar end by replacing solvated water molecules, thereby changing the solvation structure, ion transport, desolvation kinetics, and Zn deposition, while the hydrophobic side has repulsive and antifouling properties, reducing water activity at the electrode-electrolyte interface, which results in suppressed HER and side reactions for homogeneous 3D Zn plating (Figure 1). Notably, molecular materials not only assist in achieving uniform Zn2+ nucleation and deposition at the anode but also stabilize the cathode by suppressing active material dissolution, facilitate Zn2+ ion intercalation/deintercalation, and enhance reaction reversibility. Nevertheless, their functions at different locations largely rely on their functional groups, redox properties, size, and concentration.
How can adding tiny amounts of special molecules improve the stability of a zinc battery?
“Small is better and beautiful,” as is observed for single atom catalysts (SACs) which involve individual metal atoms (e.g. Pt, Fe, Ni, Co) on a supporting substrate and can offer exceptional catalytic activity and selectivity over analogous heterogeneous catalysts having bigger particle sizes. Intriguingly, trace amounts, even 5 mM concentration of additive molecules in the electrolyte have an enormous impact on changing the electrode surface, solvation structure, and breaking the intermolecular H-bonding between water molecules (chaotropic molecules), thereby, reducing water activity at the electrode-electrolyte interface and inhibiting HER. Small molecules make a screening layer on the electrode surface, creating a local lean water environment and helping to generate homogeneous electric flux and control uniform Zn deposition via promoting 3D surface diffusion, resulting in dendrite-free Zn plating/stripping. Notably, HER is associated with the number of solvated water molecules in the first sphere of Zn2+ (primary) solvation sheath; complexation with small molecules reduces the number of solvated water molecules coordinated to Zn2+, thereby, reducing the chance of decomposition of water dramatically. This lean water electrolyte, electrodes (both cathode and anode), and separators are beneficial for better performance of AZIBs. Nevertheless, water has a paradoxical effect as too little water can affect the ionic conductivity and increase the viscosity, resulting in shortening of lifetime. Additionally, decomposition of additives together with electrolytes at the electrode surface generates solid interphases (SEI for anode and CEI for cathode), which protect the electrodes from corrosion in contact with electrolytes.
One issue mentioned in the research is dendrite formation. What exactly are dendrites, and why are they dangerous for batteries?
Dendrites are needle-like Zn microstructures that grow on the anode during charging due to uncontrolled metal deposition, particularly under fast charging conditions. In AZIBs, the anode and cathode are physically separated by a porous membrane called a separator, which serves as a container for electrolyte, makes contacts, and regulates the ion transport and electric field distribution, preventing a short circuit between the electrodes and ensuring the efficient battery operation. During fast charging, rapid nucleation creates uneven deposition of Zn and tips of the uneven surface act as a charge collector for further growth, resulting a 2D dendrite (tree-like structure) growth that penetrates the separator, causing internal short circuits and battery failure, and sometimes it may even lead to thermal runaway and fire risks. A micro-short circuit is also possible from dendrites, which impairs the stable battery performance. Secondly, dendrites are not tightly bound to the Zn anode, and thus, during stripping, it can be detached and float in the form of isolated islands in the electrolyte, which results in “dead” metal deposits. This “dead” Zn may not take part in electrochemical reactions, causing capacity loss. Thirdly, owing to a higher specific surface area than a planar homogeneous Zn deposit, dendrites promote unwanted side reactions, making an unstable battery. However, the main factors of dendrite formation are concentration gradient and electric field. The onset of dendrite formation is likely related to the uneven contour of the Zn anode surface, where tiny protrusions induce the formation of tips by altering the local electric field distribution; a higher electric field and increased charge density in the tip region attract more Zn2+ ions to be preferentially deposited, leading to self-propelled rampant dendritic growth. Due to this uncontrolled nucleation and growth, local ion concentration gradients change, and large ion depletion restricts homogeneous ion diffusion and enhances dendritic growth.
How do these molecular additives help prevent corrosion and unwanted reactions in the battery?
Molecular materials having polar functional groups such as hydroxyl, carboxylate, amide, sulphonic acid, etc can chemically adsorb onto the Zn anode surface, blocking direct contact between water molecules and the anode. This surface adsorption forms a protective, water-repelling film on the Zn surface, reducing water activity at the electrode-electrolyte interface and thereby preventing corrosion, HER, and unwanted side reactions. Electrode surfaces coated with porous covalent organic frameworks (COFs) perform well, as porous COFs facilitate Zn2+ diffusion, lower charge-transfer resistance, and promote efficient desolvation by interacting with Zn2+ ion, resulting in smooth and stable plating/stripping. The carbonized coating is found to improve surface wettability and acts as an effective barrier against HER and corrosion, significantly enhancing cycling stability. While inorganic layers offer mechanical strength and stability, organic coatings provide flexibility and chemical tunability, and each alone faces inherent trade-offs. However, coating with hybrid materials is synergistically beneficial, as it combines the functional tunability of organic components with the structural stability of inorganic frameworks. Thus, by leveraging the complementary strengths of MOFs, polymers, and inorganic components, organic-inorganic hybrid coatings can offer uniform Zn2+ flux with reduced dendrite formation while providing mechanical and chemical resilience under long cycling. Furthermore, a solid electrolyte interphase (SEI) layer is formed from the decomposition of molecular additives, metal salts and solvent onto the anode surface and protects it from corrosion, thereby providing a stable electrode.
If this technology improves, where could we see these zinc batteries being used in real life? For example in renewable energy or electric devices?
The development of electrochemical energy storage, in particular, aqueous batteries, is a pressing need for providing clean and sustainable energy to societal resilience. For example, lead-acid batteries, in which aqueous H2SO4 is used as the electrolyte, have still been being used in many power sources, including starting-lighting-ignition in vehicles and household power storage for last 160 years, although they have several limitations such as low energy density (30-40 Wh kg-1), low cycle life, and toxic materials for the environment. AZIBs have unparalleled advantages over lead acid batteries, liquid flow batteries, and other aqueous metal ions (Na+, K+, Mg2+, Ca2+, and Al3+) batteries for large-scale energy storage. Advantages of AZIBs mainly stem from the Earth’s natural abundancy (around 300 times than that of Li), a highly safe aqueous-based electrolyte, and the beneficial features of non-toxic Zn metal such as high theoretical capacity of 820 mAh g-1, low redox potential (-0.76 V vs standard hydrogen electrode), and high stability in the humid and oxygen atmosphere. In the context of the sustainable energy landscape, intermittent renewable energy such as solar and wind energy are good options to store; however, these are weather dependent, e.g., wind performs well when solar does not, thus combining both could provide steadier power curves, requiring grid-scale energy storage that can bridge the gaps and supply when needed. AZIBs could be the ideal technology for grid-scale energy storage and also for wearable and flexible electrochemical energy storage devices.
Finally, what are the next big challenges or research directions scientists are working on to make zinc batteries practical for the future?
Despite the tremendous development in enhancing the AZIBs’ performance in the last couple of years, as evidenced by a plethora of publications and patents, the AZIB is not ready for practical application due to several reasons such as low energy density (30 Wh kg-1), which has to be greater than 200 Wh kg-1; poor cycle life (required: 5000 cycles at both low and high current rates: 0.2 – 2C), low Coulombic efficiency (required: over 99.99%); self-discharge rate (minimizing to less than 1% / day); low capacity retention (should be more than 80%); and operating temperature (range: -30 to 50 ⁰C). These problems originate from various components of AZIBs, such as stability and capacity of cathode, Zn anode stability, separators, and most importantly electrolytes. Holistic development of all parts can only mitigate all issues simultaneously as all parts work synergistically like interconnected gears. For example, ideal electrolyte additives can propel both cathode and anode by offering multiple functionalities such as stable electrode-electrolyte interface, interphase formation, preventing corrosion, HER, side reactions, and dendrite formation; modify solvation structure, ion transport, desolvation kinetics, and Zn deposition; and enhancing Zn/Zn2+ reversibility and capacity. Notably, it is highly imperative to focus on increasing the Coulombic efficiency of the Zn anode while applying for grid-scale energy storage. The unveiling of new multifunctional additives could be possible by utilizing machine learning (ML) coupled with generative artificial intelligence (AI), which can substantially expedite the process by minimizing trial-and -error-based experiments. Furthermore, a deep dive into understanding the charge storage and capacity fading mechanisms is extremely important through advanced characterization techniques like combinations of in situ cryo-EM, cryo-XPS, and electrochemical quartz crystal microbalance (EQCM). Additionally, since it is a new technology evolving very rapidly, the standard protocols for large-scale production have not yet been established, particularly critical parameters such as depth of discharge (DOD), state of charge (SOC), self-discharge, calendar life, loading of active mass, N/P (capacity ratio of negative to negative to positive electrode) ratio, and current density. Moreover, cost-effectiveness is of paramount importance for grid-scale energy storage, determining the viability of large-scale deployment. Finally, in light of sustainability, battery recycling possibility should be taken into account as well as biodegradable and ecofriendly materials should be prioritized as energy materials for making a circular economy with reduced environmental impact.