The world is currently facing a severe energy crisis driven by the excessive and rapid consumption and depletion of non-renewable fossil resources and the ever-increasing demand for energy due to industrialization, population growth, and urbanization. Conventional energy sources such as coal, oil, and natural gas are not only limited but also major contributors to environmental pollution and climate change. Their excessive use releases large amounts of greenhouse gases, leading to global warming, ecological imbalance, and serious health hazards.
Green hydrogen (H2) fuel has emerged as a key solution to meet the world’s growing energy demand while reducing dependence on fossil and minimizing environmental pollution. Unlike conventional H2 production methods that rely on carbon-intensive processes, green H2 is generated using renewable energy sources, making it clean, sustainable, and environmentally friendly. As the global community moves toward carbon neutrality and sustainable development, there is an urgent need for efficient and cost-effective technologies that can produce H2 without harmful emissions. However, current H2 production technologies often suffer from low efficiency, high energy consumption, and complex operational requirements, limiting their industrialization.
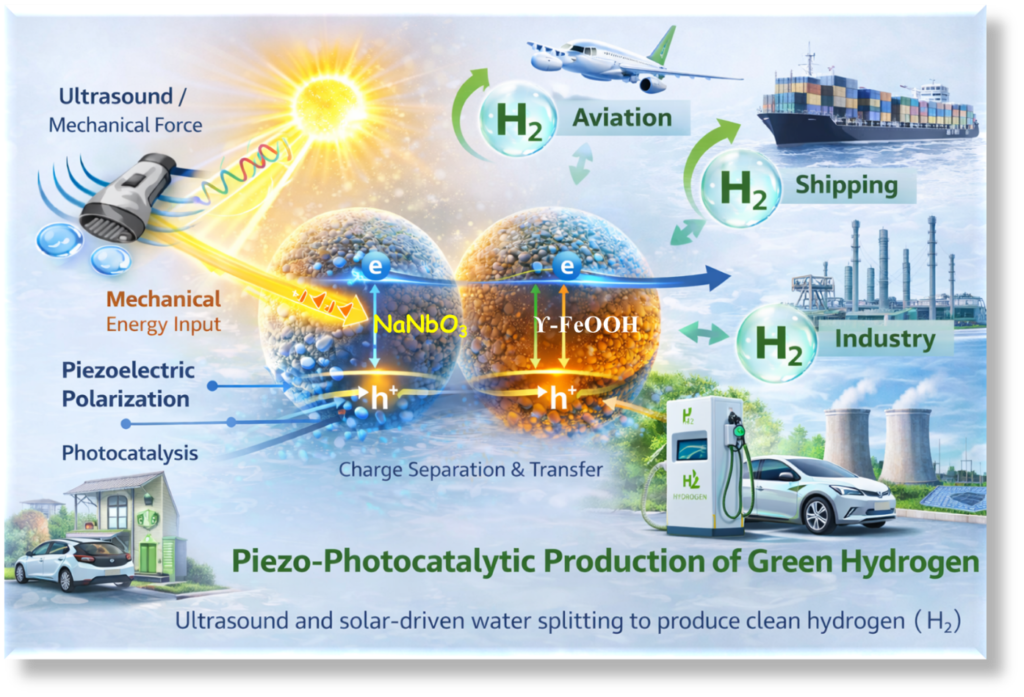
Our invention introduces a novel and sustainable approach towards sustainable H2 production using a gamma-iron oxyhydroxide (γ-FeOOH) nucleated sodium niobate (NaNbO3) binary piezophotocatalyst. This advanced material is designed to overcome the limitations of traditional catalysts by utilizing both solar energy and mechanical energy (vibrations or ultrasonic waves), to drive chemical reactions. This dual-energy harnessing strategy significantly enhances the efficiency of H2 production and offers a more practical and sustainable solution. In simple terms, when the piezocatalyst is exposed to sunlight and mechanical stress, it generates tiny charged particles known as electrons and holes. These charges are essential for initiating chemical process, particularly the splitting of water into H2 and O2. One of the major challenges in conventional systems is that these charges tend to recombine quickly, resulting in energy loss and reduced efficiency. However, in this innovation, the combination of γ-FeOOH and NaNbO3 forms a unique heterostructure that promotes efficient separation and movement of these charges. Additionally, the presence of oxygen vacancies in the material creates extra active sites and improves charge transfer, further enhancing catalytic performance.
The uniqueness of this approach lies in its piezophotocatalytic mechanism, where both light and mechanical energy work together to improve efficiency. The interaction between γ-FeOOH nanorods and NaNbO3 microcubes generates an internal electric field that facilitates effective charge separation, resulting improved H2 production compared to conventional photocatalysts. Furthermore, the synthesis process of the material is simple, low-cost, and environmentally friendly, involving hydrothermal and in-situ co-precipitation methods under mild conditions. The use of water as a solvent and the absence of toxic chemicals make the process sustainable and suitable for large-scale applications. The potential benefits of this innovation are wide-ranging. In the energy sector, it provides a clean and efficient pathway for green H2 production, which can be used as a sustainable fuel for transportation, power generation, and industrial processes. In addition, the catalyst can also be applied for environmental remediation.. By reducing reliance on fossil fuels and minimizing pollution, this technology can contribute to improved environmental quality and public health.