My research journey has been shaped by a deep interest in understanding how dynamic molecular signals govern cellular fate under stress and disease conditions. My postdoctoral journey at the University of Oxford, encompassing two successive appointments, immersed me in the fascinating realms of intracellular Ca2+ homeostasis and ADP-ribosylation biology an experience that profoundly influenced my scientific path and continues to inspire my research today. My work focuses on understanding the dynamic role of ADP-ribosylation signalling in regulating key cellular processes, particularly in the context of DNA repair, cancer biology, and immune responses. At the molecular level, I am especially interested in how Ca2+-dependent spatio-temporal regulation of ADP-ribosylation shapes cellular decision-making under conditions such as genomic instability, infection, and oncogenic transformation.
“PARP14-driven ADP-ribosylation dynamics shape the future of precision therapeutics and next-generation targeted therapies.”
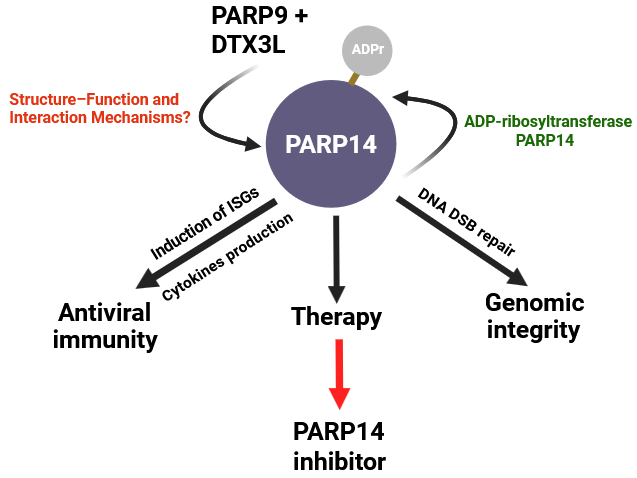
The past several decades has witnessed remarkable progress in understanding post-translational modifications as key regulators of cellular physiology. Among these, ADP-ribosylation has emerged as a highly dynamic and versatile signalling mechanism, influencing processes ranging from DNA repair to immune regulation. While early research predominantly focused on poly (ADP-ribose) polymerases such as PARP1, recent advances have brought mono-ADP-ribosyl transferases, particularly PARP14, into sharp focus. PARP14 represents a fascinating convergence of enzymatic activity and signalling complexity, as demonstrated in our recent work. Unlike classical PARPs that primarily catalyse poly-ADP-ribosylation, PARP14 functions predominantly as a mono-ADP-ribosyl transferase. This distinction is not only biochemical it reinforces a fundamentally different mode of cellular regulation. PARP14 integrates catalytic activity with multiple macrodomains, enabling it to both write and read ADP-ribose marks, thereby orchestrating highly context-dependent signalling networks.
A key aspect of our recent work has been to further elucidate the functional significance of PARP-catalysed ADP-ribosylation in cellular signalling. While ADP-ribosylation is now widely recognized as a critical regulator of diverse biological pathways, PARP-dependent mono-ADP-ribosylation remained relatively underexplored for a long time due to the lack of sensitive detection methods. By employing an improved antibody-based approach, we were able to visualize interferon (IFN)-induced ADP-ribosylation and identify PARP14 as a major enzyme responsible for this modification. Intriguingly, this signalling is reversed by the macrodomain of SARS-CoV-2 (Mac1), suggesting a potential mechanism through which viral proteins counteract host antiviral ADP-ribosylation responses. Furthermore, our findings reveal an important regulatory axis involving PARP9 and its binding partner, the E3 ubiquitin ligase DTX3L, which together modulate PARP14 activity through protein–protein interactions as well as through the hydrolytic function of the PARP9 macrodomain. Notably, we also provide the first visualization of ADP-ribosylation-dependent ubiquitylation in the interferon response, revealing a previously unrecognized layer of crosstalk between these two critical post-translational modifications. These findings not only expand our understanding of interferon-driven signalling networks but also shed light on how pathogens may evade host defence mechanisms. One of the most compelling aspects of PARP14 biology lies in its role in immune regulation. It acts as a critical modulator of cytokine signalling pathways, particularly through its interaction with transcription factors such as STAT6. By fine-tuning gene expression programs associated with inflammation and immune polarization, PARP14 contributes to the balance between protective immunity and pathological responses, positioning it at the intersection of cancer, infection, and inflammatory diseases. Beyond immunity, emerging evidence highlights the involvement of PARP14 in DNA damage responses and genome stability. Although traditionally overshadowed by PARP1 and PARP2 in this domain, PARP14 is now increasingly recognized as a contributor to repair pathway choice and replication stress responses. Its ability to modulate protein–protein interactions through ADP-ribosylation introduces an additional regulatory layer that is only beginning to be appreciated.
From a translational perspective, PARP14 represents a promising yet underexplored therapeutic target. The clinical success of PARP inhibitors in oncology has demonstrated the power of targeting ADP-ribosylation pathways. However, most current inhibitors lack specificity for mono-ADP-ribosyl transferases. The development of selective PARP14 inhibitors could therefore open new avenues for treating cancers with dysregulated immune microenvironments, as well as diseases characterized by aberrant cytokine signalling. Equally exciting is the potential integration of PARP14 biology with emerging therapeutic strategies. Nanotechnology-driven drug delivery systems may enable precise targeting of ADP-ribosylation pathways in specific tissues or cell types. In parallel, the intersection of PARP14 signalling with metabolic and epigenetic networks suggests opportunities for combination therapies that exploit multiple vulnerabilities within diseased cells.
Despite these advances, several important questions remain. What determines substrate specificity in PARP14-mediated ADP-ribosylation? How is its activity dynamically regulated across different cellular contexts? And crucially, how can we translate mechanistic insights into clinically viable interventions without disrupting physiological homeostasis? Addressing these challenges will require interdisciplinary efforts that bridge structural biology, chemical biology, and systems-level analyses. In this evolving paradigm, PARP14 stands as a compelling example of how expanding our understanding of so-called “non-canonical” signalling pathways can reshape fundamental biology and inspire innovative therapeutic strategies.
In conclusion, the study of PARP14 and mono-ADP-ribosylation is rapidly evolving from a niche area into a central theme in modern biomedical research. By unravelling its complex regulatory networks, we not only deepen our understanding of cellular signalling but also pave the way for next-generation interventions in DNA repair, oncology, as well as infection and immunity.
Looking back, I have come to realize that finding molecular mechanisms is not only an academic pursuit it is central to transforming how we approach disease biology. The path of research is often demanding and uncertain, yet each question explored, and each experiment conducted adds a meaningful step to our interpretation. As we look ahead, the ability to decode and precisely target such dynamic signalling systems will be key to developing the next generation of therapeutic strategies in cancer, infection, and immunity.