Inside every human gut live trillions of bacteria that are for the most part beneficial to our health. These microbes, collectively known as the gut microbiome, play an essential role in digestion, immunity, and overall well-being. In recent years, the gut microbiome has become a rapidly evolving area of scientific research, as we continue to discover its critical role in human health, disease, and metabolism. This is particularly important for populations with weakened immune systems, including individuals living with HIV, as well as those with altered or developing immune systems for pregnant women and their infants, respectively. We have conducted research in India on how immunocompromising conditions such as anemia and HIV may shape the gut microbiome and contribute to poor health outcomes. By studying these interactions, particularly in resource-limited settings like India, we aim to identify ways to prevent adverse health outcomes and improve the well-being of vulnerable populations with these conditions. This research is an example of a successful Indo-US collaboration of institutions and investigators. We are based out of the Department of Epidemiology at Columbia University and our other US collaborators included those from Johns Hopkins University (JHU), Cornell University and University of Maryland, Baltimore. Our Indian collaborators were from Byramjee Jeejeebhoy government medical college (BJGMC) and the BJGMC-JHU clinical research site.
“The gut microbiome is not just a collection of bacteria, it is a powerful system that shapes immunity, metabolism, and health across generations.”
In our recent work published in Nature Communications (Mandell and Wang et al., Nat Comm 17, 3097 (2026)), we analyzed data from the PRACHITi (Pregnancy Associated Changes in Tuberculosis Immunology) cohort study in Pune, India. This was a cohort of pregnant women living with and without HIV that were enrolled at BJGMC, and we followed the mothers from pregnancy through 1 year postpartum, along with their children. In this study, we sought to understand the influence of HIV infection on the gut microbiome and metabolome in mothers and their infants. Globally, millions of women of reproductive age are living with HIV, with the highest burden in resource-limited settings where there are high rates of maternal morbidity and mortality due to HIV. During pregnancy, HIV infection is associated with an increased risk of adverse birth outcomes such as preterm birth and low birth weight. In addition, children who are HIV-exposed uninfected (CHEU)-meaning they are born to mothers living with HIV but do not acquire the virus themselves-face higher risks of mortality, infections, poor growth, and developmental challenges compared to children born to mothers without HIV-children who are HIV-unexposed uninfected (CHUU). We believe that understanding how maternal HIV affects the gut microbiome of both mothers and their infants may identify specific microbes that contribute to these adverse health outcomes and could be critical to identify interventions to reduce infectious morbidity and mortality in these vulnerable populations.
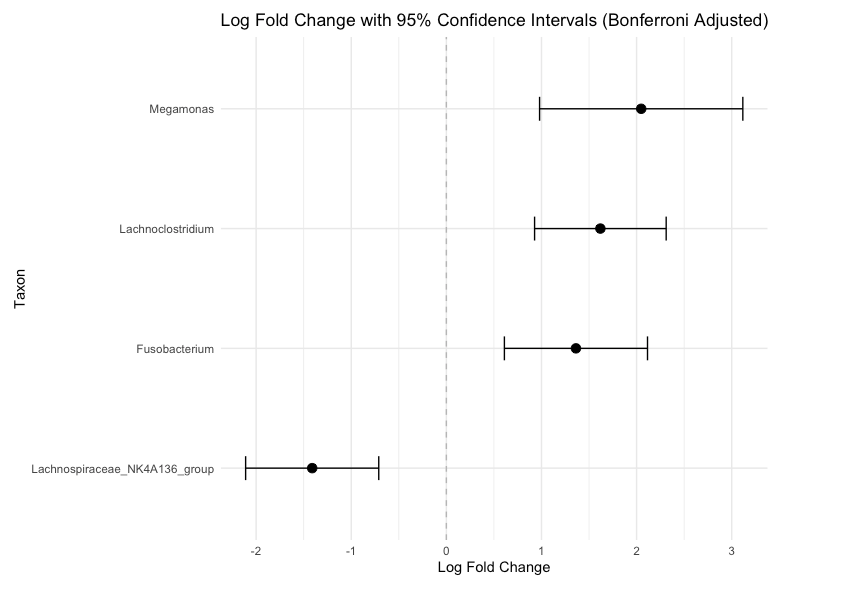
The impact of HIV infection on the gut microbiome profile is a subject of ongoing study. HIV affects not only the immune system throughout the body but also the immune cells in the gastrointestinal tract. As a result, the balance of microbes in the gut could be disrupted, a condition known as gut dysbiosis, which can trigger inflammation and metabolic changes that contribute to adverse health outcomes. These disruptions in the gut microbiome may suggest potential biological pathways through which maternal HIV infection affects both maternal and infant health. In our cohort, we observed gut dysbiosis and distinct metabolomic profiles among women with HIV (WHIV) compared with seronegative (SN) women, and their infants. Several gut bacteria were differentially abundant by HIV status, and some of these bacteria are known to increase inflammation and adverse birth outcomes such as preterm birth and low birth weight. Together, our findings suggest that changes in the microbiome profile may play an important role in shaping health outcomes and may help explain persistent disparities observed among HIV-exposed populations. The genus Fusobacterium stood out in our analysis, showing higher abundance among WHIV in their second and third trimesters. This was noteworthy to us as Fusobacterium has previously been linked to preterm birth in the oral, vaginal, and placental microbiomes, as well as to reduced immune recovery and dysfunction among individuals living with HIV. Yet, its role within the gut microbiome, especially among pregnant women with HIV, remains less clear. Its presence may represent a potential pathway linking HIV-associated microbial dysbiosis to adverse health outcomes. This raised another important question for us: how could the changes we found in the microbiota be linked to the systemic metabolome? In turn, it led us to conducting a multi-omics approach to better understand whether pathways may be shared between microbes and metabolites. We observed Fusobacterium was correlated with metabolites such as 2-hydroxyglutarate, suggesting shifts in glutamine and glutamate metabolism that may subsequently impact immunity and neurotoxicity. These results highlight the connection between the gut microbiome and metabolism, suggesting microbial and metabolic disruptions may jointly contribute to inflammation and other adverse conditions. This points to a larger role for microbiome-metabolome interactions in shaping maternal and infant health in the context of pregnancy and HIV.
Another striking finding of ours was the differential presence of bacteria from the Lachnospiraceae family in CHEU. These bacteria have been linked to promoting regulatory T-cell differentiation, a type of immune cell that suppresses the activation and proliferation of other T cells, which may limit CHEU’s ability to respond effectively to infections. They have also been associated with gut dysbiosis and immune activation in infants. These findings suggest that the presence of specific microbial communities may impact long-term health outcomes in CHEU. This population could benefit from carefully tested interventions aimed at supporting the growth of beneficial bacteria that reduce the risk of infectious morbidity and other adverse health conditions.
“Understanding how HIV alters the gut microbiome in mothers and infants can open new pathways to improve health, reduce risks, and protect future generations.”
Although research on the maternal-infant microbiome is still limited, it represents a promising and rapidly expanding field, particularly in India with its own distinct microbiome profile, with important implications for improving maternal and infant health outcomes. Pregnancy is a critical period for shaping both maternal and infant health trajectories, making it an important target for future interventions. We believe this work introduces a new direction for exploring whether approaches such as probiotics, prebiotics, or dietary modifications could improve outcomes during pregnancy. At the same time, integrating multi-omics approaches will be key to understanding how the microbiome interacts with the systematic metabolome and immune function. In countries like India, where microbiome research is a growing field, further focus on maternal-infant populations may offer new insight into underlying biological pathways for many diseases and their connection to the gut microbiome. Advancing research in this field could help inform new prevention and treatment strategies that support healthier pregnancies, improve birth outcomes, and promote early childhood development. Ultimately, these efforts have the potential to reduce the global burden of disease and mortality, particularly in resource-limited settings where maternal and infant health risks remain disproportionately high. For young researchers, the maternal-infant microbiome represents a compelling scope of research to explore. Despite recent advances, gaps remain in our understanding of how microbial, metabolic, and immune pathways interact during pregnancy and postpartum, particularly in populations affected by HIV or similar conditions. We believe some of the most important strategies young scientists must embrace to address these challenges include interdisciplinary approaches, innovative study designs, and studying diverse populations. We encourage those whose curiosity has been sparked by this research to explore this growing field. By contributing to microbiome studies, the next generation of researchers can have the opportunity to advance scientific understanding in an exciting field and help develop interventions that modify the gut microbiome and metabolome to improve health outcomes for both mothers and infants in India and around the world.