The global shift toward renewable energy is no longer a distant ambition but an unfolding reality. Solar panels and wind turbines are rapidly transforming how we generate power, yet they bring with them a persistent and consequential challenge: intermittency. The sun does not always shine, and the wind does not always blow. Bridging this gap requires efficient, safe, and genuinely sustainable energy storage systems. While much attention has been directed at improving energy storage devices such as batteries and supercapacitors, the real transformation may lie in rethinking the materials that make them work. Conventional energy storage technologies, particularly lithium-ion batteries, have powered everything from smartphones to electric vehicles. However, their environmental and ethical footprints are difficult to ignore. These systems depend on scarce and geopolitically sensitive elements such as lithium, cobalt, and nickel, whose extractions are energy-intensive, often linked to ecological degradation, and in some regions entangled with serious human rights concerns. Improper disposal further introduces toxic materials into soil and water systems. As a result, the search for greener and safer alternatives has become a central theme in modern materials science. A truly sustainable energy storage material must go beyond performance metrics such as energy density and cycle life. It should be abundant, non-toxic, recyclable, and ideally derived from renewable resources. This broader and more demanding perspective has directed researchers toward bio-derived materials, alternative chemistries, and safer electrolytes. Among these, hydrogel-based electrolytes are emerging as a particularly compelling solution.
Hydrogels are, at first glance, deceptively simple: soft, water-rich materials that resemble a sponge. At the molecular level, however, they are sophisticated three-dimensional networks of hydrophilic polymers capable of retaining large quantities of water while maintaining structural integrity. This unique combination of softness and stability imparts hydrogels with remarkable versatility and they are already widely deployed across an impressive breadth of applications. In medicine, they are used for drug delivery, tissue engineering, and wound dressing, while in diagnostics they enable sensitive biosensing. They also serve as water-retaining materials in agriculture and as adsorbents for pollutant removal in environmental applications. Beyond this, hydrogels are increasingly explored in soft robotics and wearable electronics due to their responsiveness, biocompatibility, and mechanical flexibility. Hydrogels are formed by covalent and ionic crosslinking of polymer chains, creating a three-dimensional architecture that traps water within its matrix. Covalent bonds ensure structural stability, while ionic and non-covalent interactions contribute flexibility, self-healing behaviour, and the ability to adhere to surfaces.
In every electrochemical energy device, whether a battery, a supercapacitor, or a fuel cell, the electrolyte occupies a central and often underappreciated role. It is the medium through which ions travel between electrodes. Its properties govern the device’s operating voltage window, ionic conductivity, interfacial stability, safety, and long-term durability. For decades, this critical component has been dominated by liquid systems: aqueous solutions, organic solutions, and ionic liquids. While effective, these conventional electrolytes carry several serious liabilities. Organic electrolytes are flammable and toxic; ionic liquids are expensive; aqueous systems have low voltage window. This necessitates safer and affordable alternatives.
Hydrogels offer a compelling resolution to the limitations of conventional electrolytes. In the context of energy storage, they combine the best features of liquid and solid systems. Like liquids, they enable efficient ion transport through their water-rich interconnected pore networks. Like solids, they provide mechanical stability, eliminate leakage, and enable compact as well as flexible device architectures. Hydrogel electrolytes can be synthesized and operated under ambient conditions, making them cost-effective and user-friendly.
A wide range of biopolymers are available for hydrogel electrolyte design. Cellulose offers excellent mechanical strength and stability, while chitosan provides a positively charged network that supports efficient ionic conductivity. Alginate and gelatin contribute flexibility and strong film-forming ability. Other abundant materials such as guar gum and carrageenan further expand the scope for sustainable electrolyte development. What makes these materials scientifically powerful is the diversity of crosslinking strategies available. Covalent crosslinking, using agents namely glutaraldehyde and glyoxal, produces dense mechanically robust networks but may involve toxic reagents and can restrict ion mobility. Ionic crosslinking, by contrast, relies on electrostatic interactions between charged polymer groups and counterions from a salt or crosslinker. This approach is simpler, greener, and often preserves or enhances ionic conductivity, since the crosslinker ions themselves become mobile charge carriers within the hydrogel. Several hydrogel electrolytes have exhibited outstanding performance in electric double layer capacitors. A hybrid aqueous electrolyte system prepared using sulfuric acid and sodium sulfate with tungsten oxide electrodes demonstrated an exceptional cycle life of 250,000 cycles, representing one of the most durable systems. Similarly, chemically cross-linked polyacrylamide organo-hydrogel-based system achieved a long cycle stability of 100,000 cycles, while azepanium-based ionic liquid systems paired with activated carbon delivered 55,000 cycles. In our recent research, a series of biopolymer-based hydrogel electrolytes demonstrated progressively improved performance. The chitosan-sodium thiosulfate system exhibited the highest reported cycle life of 1,58,924 cycles, along with a high areal capacitance of 505.5 mF cm-2. This was followed by the gelatin-Al3+ system, which delivered 83,000 cycles. These results clearly demonstrated that systematic tuning of biopolymer chemistry and ionic crosslinking could lead to significant improvements in both electrochemical performance and long-term durability.
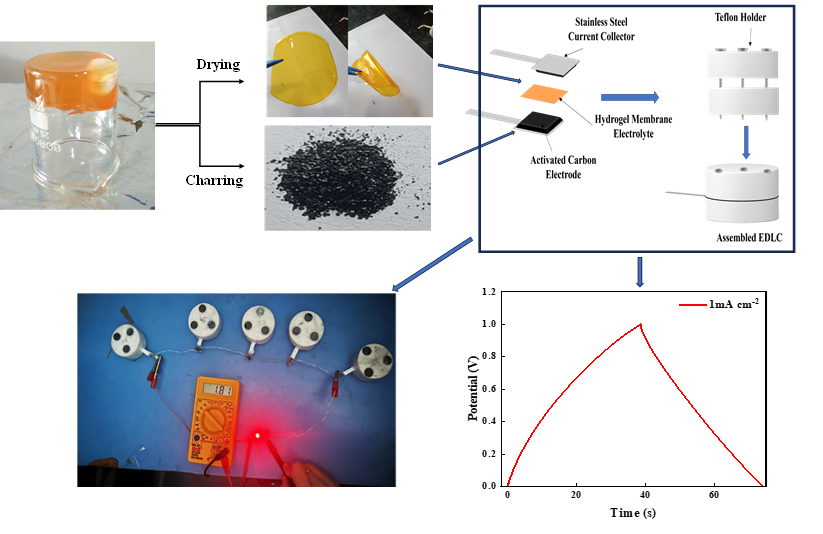
The hydrogel, shown in the inverted beaker, was synthesized by covalent crosslinking of gelatin with glutaraldehyde and subsequently processed into a flexible electrolyte film. Activated carbon powder was also derived from the same hydrogel through a carbonisation process. This was followed by integration with carbon-based electrodes to assemble an electric double layer capacitor. The fabricated cells were then connected in series to demonstrate practical applicability, successfully powering a red LED. This representation illustrates simple fabrication and ability of the hydrogel electrolytes to deliver usable electrical output for real-world applications.
Despite the significant advancements outlined here, serious challenges remain on the path from laboratory discovery to practical deployment. The electrochemical stability window of hydrogel electrolytes typically limited to low value. Strategies to widen this window include the use of water-in-salt electrolytes at high salt concentrations. Self-healing hydrogels, which automatically repair mechanical damage, represent another exciting frontier, offering the prospect of dramatically extended device lifetimes. Large-scale production of hydrogel membrane electrolytes is another challenge that can be addressed through innovative engineering. Finally, integrating hydrogel electrolytes with advanced electrodes offers promising near-term performance breakthroughs. For early-career researchers interested in the intersection of polymer chemistry, materials science, and electrochemical engineering, this field presents an exceptional opportunity. The challenges are significant, and materials developed today will shape future energy storage. Moreover, the barriers to entry are lower than commonly assumed. Biopolymers are inexpensive and widely available, while straightforward fabrication approaches make experimentation accessible even in modest laboratory settings. As the discipline advances, there is a growing recognition that achieving high-performance energy storage must go hand in hand with sustainability. The next generation of researchers will play a crucial role in advancing this balance, contributing to technologies that are both efficient and sustainable.