Nanozymes: From Enzyme Mimicry to Real-World Applications: The groundbreaking discovery of intrinsic peroxidase activity (POD) for ferromagnetic nanoparticles reported in Nature Nanotechnology has led to a huge increase in research on artificial nanozymes over the past few decades. However, a fundamental question remains: How did the concept of “nanozymes” gain popularity in the scientific community, and what is the underlying mechanism that enables these tiny nanozymes to replicate the catalytic behaviour of natural enzymes? The term “nanozymes” refers to nanomaterials that mimic the catalytic function of natural enzymes, such as Horseradish peroxidase, by providing a high surface-active site, including a metal centre, a defect, or edge sites, where the substrate can adsorb and undergo catalytic transformation. Similar to the natural enzymes, these artificial enzymes primarily operate through an electron transfer mechanism. Artificial nanozymes, particularly those on metal-based surfaces like Fe, Cu, or Ni, facilitate redox reaction by undergoing a reversible change in oxidation state (e.g., Fe2+/Fe3+, Cu+/Cu2+), therefore enabling electron transfer efficiently. This reversible redox behaviour triggers their enzyme-mimicking activity, allowing their use as a catalyze the reactions such as H2O2 decomposition, generation of reactive oxygen species. These highly reactive intermediates are unstable and subsequently oxidize the target substrate, such as TMB/OPD. The oxidation of these substrates can be monitored by observing the color change of the oxidized product. This colorimetric sensing strategy serves as the foundation for the detection of a wide range of biomolecules and heavy metals. The catalytic efficiency of these nanozymes in comparison to natural enzymes can be quantitatively measured using the Michaelis-Menten kinetics. The key Michaelis-Menten parameters, such as Michaelis constant (Km), maximum reaction velocity (Vmax), and turnover number (Kcat), provide insight into substrate affinity, catalytic capacity, and overall enzymatic performance. Artificial nanozymes offer the advantage of tunability of their physicochemical properties, such as size, shape, and composition, enabling optimization of their catalytic performance. Although these artificial nanozymes generally offer lower substrate specificity than natural enzymes, they offer significant advantages such as superior stability, cost-effectiveness, and the ability to work under harsh conditions. These advantages make them a promising candidate for a wide range of applications, including chemical sensing, environmental remediation, and biomedicine.
“Nanozymes show that tiny materials can perform powerful chemistry mimicking nature while working stronger, faster, and in tougher conditions.”
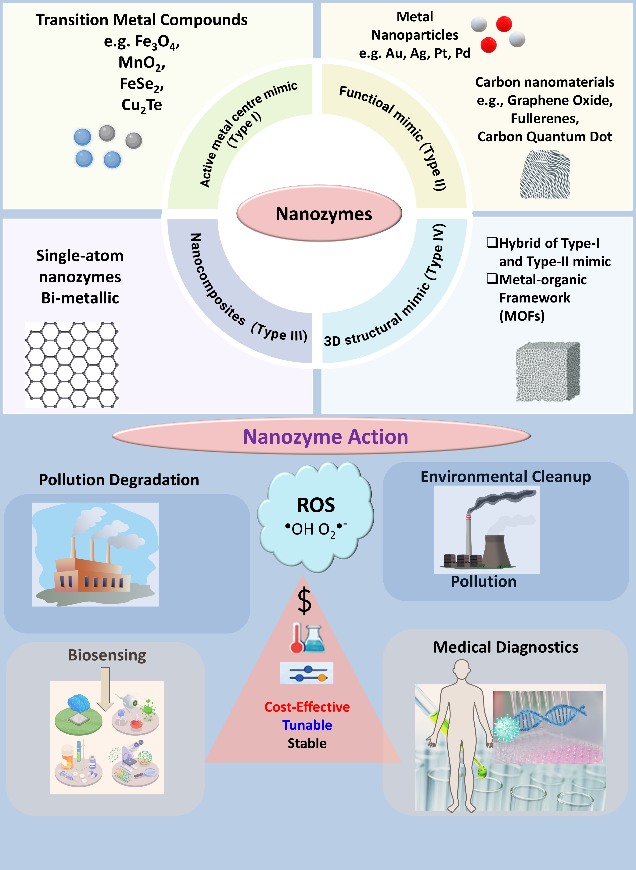
Types of Nanozymes: The nanozymes can basically be categorized into four classes based on their enzyme-mimicking behaviour.
(I). Type I nanozymes, termed as “active site centre mimic”, include transition metal compounds such as Fe3O4, MnO2, CuSe2, FeSe2, Cu2Te, etc., which imitate natural enzyme active sites.
(II). Type II, known as “Functional mimic,” consists of metal nanoparticles (e.g., Au, Ag, Pt, Pd, Ir) and carbon-based nanomaterials like graphene oxide, fullerene, and carbon quantum dots.
(III). Type III nanozymes are “nanocomposites” combining the features of both Type I and Type II materials, including hybrid and metal-organic framework (MOFs), to enhance catalytic performance.
(IV). Finally, type IV nanozymes are “3D structural mimics,” such as single-atom nanozymes and bimetallic alloys with etched channels, designed to replicate the structural complexity and efficiency of natural enzymes.
Fe, V, metal, and MOF-based nanomaterials exhibit POD; Cu, Mo, and Pt-based nanomaterials act as oxidase mimics; metal, metal oxide, Pt, and Pd-based nanomaterials function as catalase mimics; carbon, Ce, and melanin-based nanomaterials also showed catalase-like mimic behavior; while carbon-based, monolayer-functionalized gold nanoparticle, and MOF-based nanomaterials mimic hydrolase activity.
Our group focuses on the rational design and synthesis of metal-based nanozymes and 2D material–doped metal nanozymes that effectively mimic the catalytic activity of peroxidase enzymes, enabling enhanced reactive oxygen species generation and efficient catalytic degradation process. We have developed Cu2Te, FeSe2, and their respective composite such as Cu2Te/rGO and FeSe2/rGO, which exhibited strong peroxidase and catalytic-like behaviour. We have also shown that the combination of Cu2Te/rGO or FeSe2/rGO showed enhanced POD as compared to their pristine Cu2Te or FeSe2 NPs. The catalytic mechanism of these nanohybrids can be generalized as a synergistic nanozymes system for substrate oxidation, particularly for substrates such as TMB and OPD. In this system, the metal in the pristine component undergoes a redox cycle, which activates H2O2 through a Fenton-like pathway, to generate highly reactive •OH radicals, primarily responsible for oxidation. The presence of a reducing environment facilitates the formation of Cu+, thereby enhancing •OH radicals’ generation and catalytic turnover. Simultaneously, the rGO matrix plays a significant role for the enhance POD catalytic activity by accelerating electron transfer and stabilizing the intermediate state. The •OH radical generated during the reaction oxidizes the TMB/OPD substrate into its respective oxidized product, leading to visible colorimetric detection. Additionally, pristine nanoparticles are uniformly dispersed over the rGO surface owing to the strong interfacial interaction, thereby increasing the accessibility of more active sites and facilitating efficient substrate adsorption in the proximity to the catalytic centre. Overall, the synergistic combination between redox-active pristine nanoparticles and conducting rGO surface results in the efficient electron transfer, increased ROS generation, and superior POD activity via the oxidation of TMB/OPD.
Furthermore, In our study, molecular docking analysis of the FeSe2/rGO nanohybrid revealed significantly enhanced binding affinities toward peroxidase-related proteins, including 1HRP (−9.9 kcal/mol), 1PRX (−11.3 kcal/mol), 2BOQ (−10.5 kcal/mol), 4W7J (−10.7 kcal/mol), 6IU1 (−11.9 kcal/mol), 6QZO (−10.2 kcal/mol), and 7QYO (−11.0 kcal/mol), compared to pristine FeSe2 nanoparticles, confirming stronger interactions and superior catalytic potential of the nanohybrid. Future Prospective: The unique architecture of the nanomaterial contributes to the intrinsic POD catalytic behaviour, which can open up new doors for the development of a nanozyme-based platform for sensing, environmental, and biomedical applications. There are still some drawbacks, such as reproducibility, recyclability, specificity, and selectivity. These nanomaterial/nanohybrid can be functionalized/turned based on the selectivity and colorimetric biosensors for the detection of cysteine, glucose, H2O2, heavy metals, and cholesterol, etc. Integrating such nanozymes into portable sensing devices, paper-based strips, and microfluidic systems could enable them for real-time and on-site detection. One can study the underlying mechanism using advanced spectroscopic and computational studies, which can provide more details regarding the electron transfer process and active site behaviour. Artificial intelligence and machine learning could play a vital role in screening the selectivity towards a particular target. Furthermore, one can study the biocompatibility/stability experiment, which can open up new doors in the biomedical area, such as diagnostic, imaging, and therapeutic applications, including ROS-mediated antibacterial or anticancer strategies. Overall, this work lays a strong foundation for the scalable development of cost-effective, robust, and multifunctional nanozyme systems with board interdisciplinary approach. Thus, nanozymes offer a unique platform that integrates enzymatic efficiency with material stability, paving the way for the next-generation catalytic system.