When we think about chemical sensing, we often imagine a device that gives a digital readout or a test strip that changes color. But chemistry is now moving toward something far more elegant: materials that can respond to their surroundings by changing the way they emit light. In the recent Communications Chemistry article, “A state-switchable TADF macrocycle for multi-analyte sensing and hydrogen gas-driven emission enhancement,” researchers describe exactly such a system: a smart molecular material that can alter its glowing behavior depending on which chemical species it encounters.
“This smart molecule doesn’t just glow it changes its light to ‘tell’ us what chemical is present, turning light into a language of detection.”
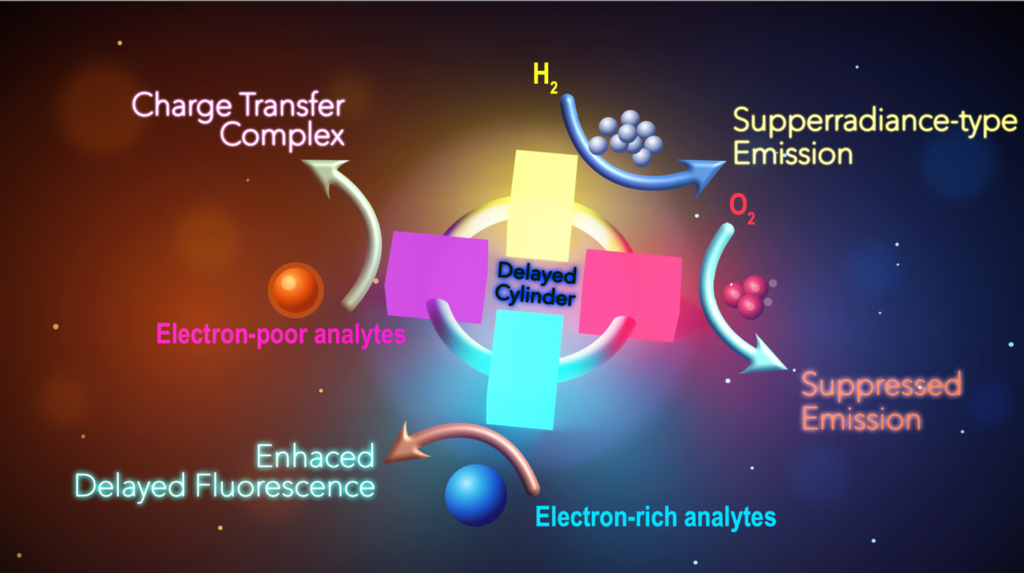
What makes this work especially exciting is that the material does not simply become brighter or dimmer in a routine way. It can switch between different emission states. That means its light output is not fixed. Instead, it is adaptive, almost conversational. Different chemical guests push the molecule into different photophysical modes, and those changes can be read out through shifts in brightness, color, and delayed light emission. In a world that increasingly needs fast, sensitive, and selective detection technologies, this is a powerful idea. The central material in the study is called CPCQ. It is a macrocycle, meaning a large cylindrical-shaped molecule, built from four twisted donor–acceptor units arranged in a cyclic architecture. That ring-like structure matters. In many light-emitting molecules, the chromophore, the light-producing part, is treated as a more or less fixed object. Here, the cyclic design creates a cooperative platform in which multiple emitting units can work together. The result is a material with strong baseline performance even before sensing begins: it shows a photoluminescence quantum yield of 78%, a delayed lifetime of 243 nanoseconds, and a reverse intersystem crossing rate of 2.68 × 10⁷ s⁻¹. In plain terms, it is already an efficient glow-emitter, and it is unusually good at managing excited states in a controlled way.
To understand why this matters, it helps to unpack one key term: TADF or thermally activated delayed fluorescence. In ordinary fluorescence, a molecule absorbs energy and quickly emits light. In TADF materials, some of that energy temporarily enters a darker triplet state before being thermally recycled back into a light-emitting singlet state. That recycling can improve efficiency because energy that might otherwise be lost is recovered as light. TADF has already become important in advanced display technology and organic light-emitting devices. What this paper shows is that TADF can also be turned into a sensing strategy.
That is the real conceptual leap. Most sensors are designed to recognize one target and give one kind of signal. CPCQ behaves more like a photonic decision-maker. Electron-deficient guests cause it to form exciplexes, which reduce fluorescence and shift the emission. Electron-rich guests do something different: they enhance delayed fluorescence through what chemists call the heavy atom effect. Oxygen produces yet another response by quenching charge-transfer emission and restoring a blue-shifted locally excited state. Then comes the most striking result of all: hydrogen not only changes the emission state but boosts the locally excited emission dramatically, giving a radiative rate of 2.07 × 10⁸ s⁻¹ and a photoluminescence quantum yield of 93%.
This matters because each of these chemicals leaves a distinct optical fingerprint. The material does not merely say “something is present.” It says, in effect, “this type of guest is here, and I will respond in this particular way.” That is what scientists mean by multi-analyte sensing. A single material can detect multiple classes of chemicals by producing different optical outputs. For practical applications, that is a major advantage. Instead of building separate sensors for each target, one can imagine integrated sensing platforms based on one adaptable molecular system.
Hydrogen sensing deserves special attention. Hydrogen is widely discussed as a clean energy carrier, but it is also highly flammable and difficult to detect because it is colorless and odorless. Reliable hydrogen sensing is therefore essential in storage, transport, and fuel technologies. In this study, hydrogen triggers a particularly strong enhancement in emission rather than simple quenching. That is useful because a signal that becomes brighter can be easier to detect against background noise than one that merely fades. A molecule that turns on more strongly in the presence of hydrogen could therefore be valuable in safety monitoring and next-generation energy systems.
Another notable aspect of the paper is its suggestion of superradiance-like behavior. The authors propose that the dramatic increase in locally excited emission under hydrogen may arise from cooperative action among the donor–acceptor units in the macrocycle. For a general reader, the important point is this: the molecule may be doing more than the sum of its parts. Its architecture allows several subunits to act together, producing a response that is amplified beyond what one might expect from a single isolated emitter. Even if future studies refine the mechanism, the broader lesson is already clear: smart function can emerge from collective molecular design. This work offers an important message about modern chemistry. Today’s functional materials are rarely just “molecules” in the old static sense. They are information-rich systems. Their shapes, electronic structures, excited states, and host–guest interactions are all engineered together. This is chemistry meeting physics, materials science, and engineering in a single design. The best new molecules are not simply stable or beautiful; they are responsive, programmable, and purposeful. This study is also a compelling teaching example. It links textbook ideas such as fluorescence, excited states, charge transfer, host–guest chemistry, and molecular architecture to a real application with societal relevance. This highlights why investment in fundamental molecular research matters. A discovery that begins with an unusual light-emitting macrocycle can lead to practical tools for environmental monitoring, industrial safety, energy infrastructure, and optoelectronic devices. The pathway from basic science to application is often nonlinear, but this paper shows how quickly that bridge can become visible.
“From sensing hydrogen to identifying multiple chemicals, this material shows how molecules can move from passive emitters to active, intelligent sensors.”
In my view, the most important contribution of this work is not only that it presents a new sensor. It presents a new design philosophy. Instead of asking a material to emit light in one optimized way, the researchers have built a system that can switch, adapt, and communicate through light. That is a profound shift. It moves us from passive luminescent materials to active photonic materials that respond intelligently to chemical surroundings. As chemistry advances, we will need more materials like this: compact, efficient, selective, and capable of reading the world around them through changes in light. CPCQ points in that direction. It shows that a carefully designed molecular ring can become a dynamic optical platform, sensitive to different analytes and especially promising for hydrogen detection. A smart glow may sound poetic, but in this case it is also practical science. And that is what makes this work so memorable: it turns molecular light into useful chemical language.