What first inspired you to design and study bent-core Schiff base ligands and their nickel complexes, and how did this research begin?
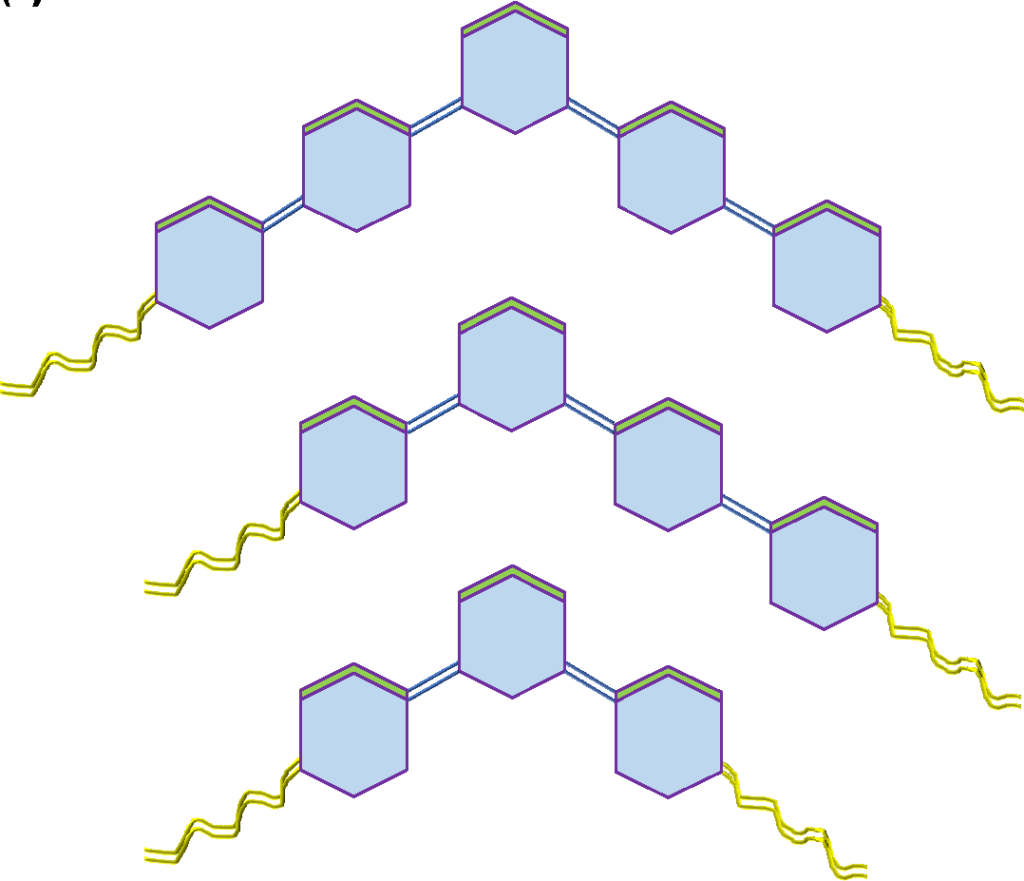
The concept of bent-core (or bent-shaped) molecular design (Figure 1) originates from liquid crystal research, where molecular shape plays a decisive role in determining mesomorphic behavior. A pioneering study by Hideo Takezoe and his group at Tokyo Institute of Technology demonstrated that non-linear, banana-shaped molecules can exhibit liquid crystalline phases, expanding the scope beyond conventional rod-like mesogens. This discovery significantly accelerated research in bent-core liquid crystals (BLCs). In India, Nandiraju V. S. Rao and his research group at Assam University, Silchar developed a unique class of asymmetrical bent-core systems, characterized by an uneven distribution of phenyl rings around a 1,3-disubstituted central aromatic core. These systems typically consist of four phenyl rings and exhibit bend angles in the range of 140°–160°, and are commonly referred to as short bent-core systems. As a member of this research group, work has been carried out on designing even more compact architectures, including three-ring bent-core molecules, which represent some of the shortest systems in this category. In addition, efforts have been made to synthesize metal complexes of such ligands with transition metals like copper, zinc, and iron to investigate how the bent molecular framework influences coordination behavior and whether liquid crystalline properties are retained or new functionalities emerge. Recently, a Ni(II) complex based on a very short bent-core ligand has been successfully developed, showing interesting structural and physicochemical properties.
Can you explain in simple terms what makes this bent-core nickel complex special and different from other metal complexes?
The shape of the ligand is bent which is not typical as other common ligands. And due the bent geometry of the ligand molecules it is not easy to form complexes. But at the same due the bent shape the electronic polarizability and electrochemical properties are different as compared to typical longitudinal or flat ligand.
Your study explores DNA and protein binding why is this important for chemistry, biology, and possible medicinal applications?
Studying the binding of metal-based complexes with DNA and proteins is important in chemistry, biology, and medicine because it provides insight into their structural and electronic behavior, helping in the rational design of stable and functional compounds. Biologically, interactions with essential biomolecules like DNA and serum albumins (HSA/BSA) reveal how these complexes are transported, distributed, and recognized within living systems. From a medicinal perspective, DNA binding is especially significant since many anticancer drugs act by targeting DNA to inhibit cell growth, while protein binding enhances drug solubility, reduces toxicity, and improves delivery through the bloodstream. Overall, understanding these interactions is crucial for developing effective, selective, and safer therapeutic agents.
What surprised you the most during the structural, computational, or biomolecular studies of this complex?
The most surprising observation from this study was the strong and sensitive biomolecular interaction behavior of the Ni(II) complex despite its relatively simple square-planar structure. In particular, the complex exhibited very low detection limits (0.075–0.188 µM) toward DNA, BSA, and HSA along with a clear fluorescence red-shift response, indicating highly efficient biomolecular recognition. Additionally, the significant binding affinity with BSA (−8.52 kcal/mol) revealed through molecular docking, supported by multiple non-covalent interactions, was notably high. From a computational perspective, the presence of a moderate HOMO–LUMO gap (4.00 eV) combined with strong ligand-to-metal charge transfer was also striking, as it reflects a rare balance between stability and reactivity. Altogether, the combination of strong binding, high sensitivity, and favorable electronic properties was an unexpected and noteworthy outcome of the study.
Do you think this type of molecule could have future applications in sensing, drug design, or biomedical research?
Indeed, this type of molecule is highly promising for future applications across sensing, drug design, and biomedical research. Its ability to produce distinct and measurable fluorescence responses upon interaction with biomolecules makes it suitable for developing sensitive detection platforms. At the same time, the strong and stable binding with DNA and serum proteins highlights its relevance in therapeutic development, particularly for designing agents that can effectively target and transport within biological systems. Furthermore, its favorable electronic structure and stability support its functionality in complex biological environments. Collectively, these properties suggest that such complexes could play an important role in next-generation diagnostic tools, targeted drug systems, and bio-related research technologies.
What are the next steps, and what still needs to be studied before we fully understand the practical potential of this system?
Although the study reveals promising structural, electronic, and biomolecular properties of the Ni(II) complex, several further investigations are required to fully understand its practical potential. Detailed biological studies, including in vitro cytotoxicity, selectivity, and mechanism of action, are necessary to confirm its therapeutic relevance, followed by in vivo evaluations to assess pharmacokinetics, bioavailability, and toxicity under physiological conditions. More advanced biophysical techniques are also needed to precisely determine the mode of DNA and protein binding beyond preliminary fluorescence and docking results. In addition, computational studies such as molecular dynamics simulations would provide deeper insight into stability in biological environments, while systematic structure–activity relationship modifications could help optimize performance. Finally, for sensing applications, parameters like selectivity, interference, and real-sample applicability must be evaluated, making these combined efforts essential to translate this system into effective biomedical or diagnostic use.