What first inspired you to explore PET plastic waste as a source for developing biomedical materials, and how did this research begin?
The inspiration for this work arose from addressing two major global challenges: the growing accumulation of plastic waste and the need for advanced, affordable biomedical materials. As a researcher in polymeric biomaterials, I have always aimed to develop functional systems that directly benefit human health. At the same time, the environmental impact of non-biodegradable plastics like PET, which persist for decades, prompted us to rethink waste as a resource rather than a problem.
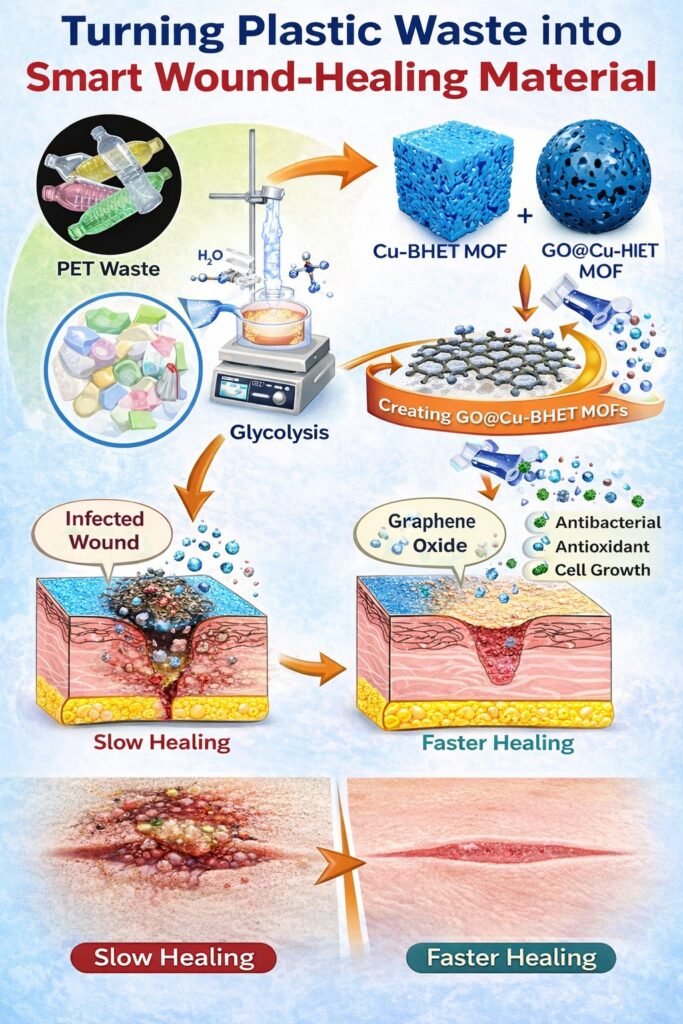
Our approach began with chemical recycling of PET through glycolysis, producing bis(2-hydroxyethyl) terephthalate (BHET). Interestingly, BHET closely resembles organic linkers used in metal–organic frameworks (MOFs), which led us to explore its potential in constructing functional materials. This marked the starting point of our research.
We then developed a copper-based BHET MOF, leveraging the inherent antimicrobial properties of copper. The breakthrough came with the integration of graphene oxide, which significantly enhanced biological performance due to its unique surface and antibacterial characteristics.
Ultimately, this work reflects a simple yet impactful idea: transforming plastic waste into a high-value biomedical material. It represents our broader vision of merging sustainability with advanced materials science to create solutions for both environmental and healthcare challenges.
Can you explain in simple terms how discarded plastic bottles were converted into a useful wound-healing material?
At first glance, a discarded plastic bottle and a wound-healing material may seem unrelated, but chemistry allows us to bridge this gap. We began with used PET plastic bottles, which were cleaned, cut into small pieces, and subjected to a recycling process called glycolysis. This process breaks the long plastic chains into smaller molecules, producing a key compound known as bis(2-hydroxyethyl) terephthalate (BHET).
BHET serves as a building block for advanced materials called metal–organic frameworks (MOFs). By combining BHET with copper ions, we created a porous, highly structured material. This structure is important because it provides a large surface area, enabling effective interaction with biological systems. Copper further contributes by offering natural antibacterial properties, helping to prevent infection.
To enhance performance, we incorporated graphene oxide, a carbon-based nanomaterial known for improving cell interaction and antibacterial activity. The resulting composite material can reduce oxidative stress, inhibit bacterial growth, and promote cell regeneration.
In simple terms, we transformed plastic waste into a smart material that supports faster wound healing. This approach not only helps manage plastic pollution but also advances the development of innovative and functional healthcare materials [1].
Your study combines sustainability with healthcare, why is this kind of waste-to-biomedicine approach important for science and society?
Integrating sustainability with healthcare offers a powerful solution to two major global challenges: plastic pollution and the need for advanced, affordable medical materials. PET waste, which accumulates in massive quantities and persists for decades, poses a serious environmental threat. At the same time, healthcare systems require materials that are not only effective but also safe, scalable, and cost-efficient. A waste-to-biomedicine approach connects these needs by transforming environmental waste into valuable biomedical resources.
From a scientific perspective, this strategy encourages innovation by treating waste as a chemical feedstock rather than a disposal problem. Converting PET into functional materials such as metal–organic frameworks enables the design of bioactive systems with antibacterial, antioxidant, and tissue-compatible properties. This expands the scope of materials science while supporting circular economy principles.
From a societal standpoint, such approaches can lower the cost of healthcare by reducing dependence on expensive raw materials. This is particularly beneficial in resource-limited settings, where affordable wound care solutions are crucial. At the same time, it helps reduce plastic accumulation and promotes high-value recycling.
Overall, waste-to-biomedicine represents a paradigm shift, demonstrating that environmental sustainability and healthcare innovation can work together to create meaningful impact.
What surprised you the most during the development or testing of the GO@Cu-BHET MOF material?
What surprised us most was the extent to which graphene oxide enhanced the overall performance, even at very low concentrations. While some improvement was expected due to its known properties, the magnitude of enhancement—particularly in biological activity—was far beyond our initial expectations.
The most striking result came from the wound healing assay, where GO@Cu-BHET MOF achieved nearly complete wound closure within 24 hours at just 10 µg/mL. In contrast, the Cu-BHET MOF without graphene oxide showed only partial healing under the same conditions. This highlighted a strong synergistic interaction between graphene oxide and the copper-based framework.
We also observed a significant boost in antioxidant activity. While Cu-BHET MOF showed moderate scavenging, the GO-integrated system reached around 80% activity at higher concentrations, indicating a more active role of graphene oxide in radical neutralization.
Importantly, these enhancements did not compromise cytocompatibility, with both materials maintaining over 90% cell viability. Overall, the synergy between components was the most unexpected and impactful finding of this study.
Do you think this material could have broader applications in wound care, infection control, or other biomedical fields?
Yes, we believe this material has strong potential beyond wound healing. The GO@Cu-BHET MOF combines key properties such as antioxidant activity, antibacterial performance, and cytocompatibility, making it suitable for a wide range of biomedical applications.
In wound care, it can be developed into advanced formats like hydrogels, coatings, or dressings that provide sustained antibacterial protection while promoting tissue regeneration. Its ability to achieve rapid healing at low concentrations makes it particularly promising for chronic wounds, burns, and diabetic ulcers.
From an infection control perspective, the material could be used as an antimicrobial coating on medical devices and implants. The combination of controlled copper ion release and graphene oxide-induced membrane disruption offers a dual mechanism to prevent bacterial growth and biofilm formation.
Additionally, its porous structure enables drug loading and controlled release, making it useful for drug delivery and tissue engineering applications. Overall, its multifunctional and tunable nature makes it a versatile platform for next-generation biomedical technologies.
What are the next steps, and what still needs to be studied before this material can move closer to real-world medical use?
While our findings are encouraging, several steps are needed before this material can be translated into real-world medical use. The immediate priority is to evaluate its performance in in vivo wound healing models, which will help confirm its efficacy, safety, and behaviour under physiological conditions. These studies will also provide insights into immune responses and tissue regeneration processes.
Another key requirement is assessing long-term biocompatibility and toxicity. Although in vitro results showed excellent cell viability, it is essential to study biodegradation, clearance, and the long-term effects of copper ion release to ensure safety.
We also aim to develop practical formulations, such as incorporating the material into hydrogels, films, or nanofiber dressings. Factors like stability, sterilization, and shelf-life must be optimized for clinical application.
From a translational perspective, ensuring scalable and reproducible synthesis is critical, especially when using PET waste as a starting material. Consistency in raw material quality must be maintained. Finally, regulatory approval will require comprehensive evaluation of safety, efficacy, and comparison with existing treatments. Overall, advancing this material toward clinical use will require coordinated efforts across materials science, biology, and medicine.