What first inspired you to investigate the molecular biology of endometriosis, and how did this research begin?
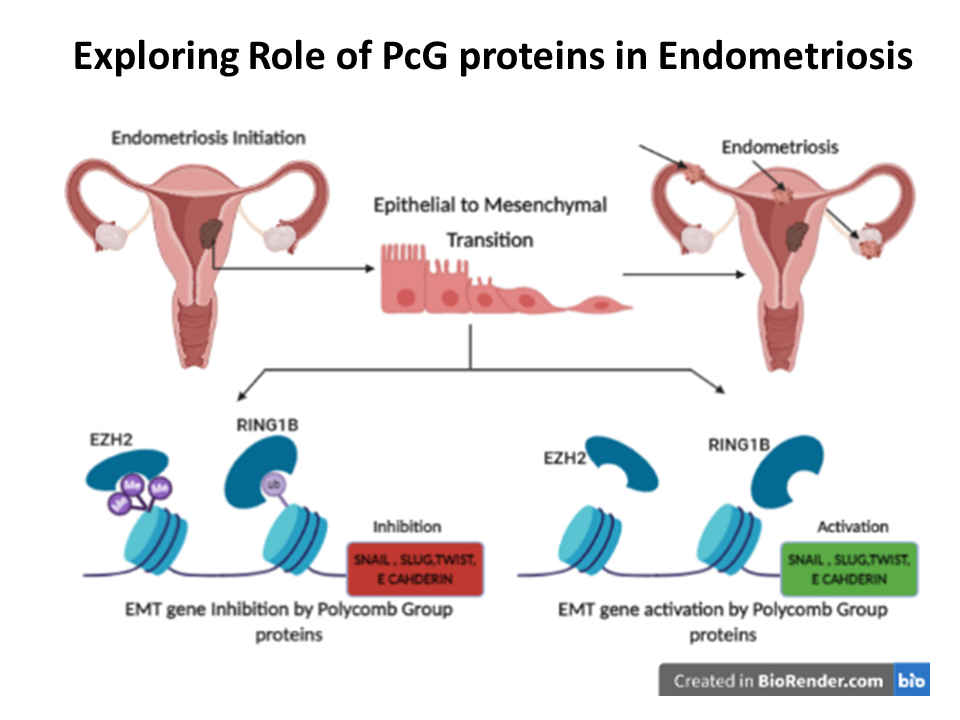
When I joined Symbiosis Centre for Stem Cell Research (SCSCR) in 2019, we wanted to study a gynecological disorder common in India but poorly understood. While many researchers study endometriosis, few focus on its epigenetic mechanisms. I noticed no one had explored the role of Polycomb repressive complex 1 in Epithelial to Mesenchymal Transition (EMT) gene regulation, so we began pilot experiments and later secured funding to pursue this. I did some pilot experiments to see if we can culture cells from endometrial biopsy as well as extract RNA and protein for molecular biology assays. We first wrote up a research proposal and submitted it to funding agency, and when the proposal was approved for funding we began work in 2024.
Can you explain in simple terms what endometriosis is and why understanding cell behavior is important in this condition?
Endometriosis is a complex disease since many researchers now believe that this disease may not be just one disease but could be 3 to 4 sub conditions, like how in early 20th century mental illness could mean depression, hysteria, bi polar disorder, schizophrenia, dementia. So far what we can say confidently about endometriosis is that it a condition that primarily affects women of reproductive age, where small bits of endometrial tissue are found outside the uterus such as ovary. Once at these sites outside uterus , the tissue leads to painful periods, painful intercourse and heavy menstrual bleeding, and may possibly lead to infertility. Thus, one of the critical aspects of endometriosis is migration of endometrial cells towards other sites and understanding it might help us in treating endometriosis.
Your study links Polycomb proteins with EMT-related genes why is this finding important for understanding endometriosis and women’s health?
Worldwide there are millions of women suffer from endometriosis, but unfortunately there is no definitive cure for this condition. Our study showed that EMT gene are active in the ectopic tissue (endometrial tissue found in lesions away from the endometrium), and we showed correlation between Polycomb group (PcG) proteins and EMT genes. If future studies confirm this correlation, then the PcG proteins could serve as potential targets for synthetic compounds that are already being used in several clinical trials for treating several types of cancers.
What surprised you the most during your research or while comparing eutopic and ectopic tissues?
When we compared the ectopic and eutopic tissue we were surprised to see the differences, I assumed there would be only minor differences, but there were major differences in expression of genes such as – crystallin gamma D (CRYGD), Follicle stimulating hormone receptor (FSHR), Diencephalon/mesencephalon homeobox 1 (DMBX1), IZUMO1R Aristaless-related homeobox (ARX1) and Leptin (LEP) to name a few. Some of these are not know to be required for endometrium but are associated with other tissues such as eyes, brain and fat cells. We also were taken by surprise when results for some markers were completely opposite in transcriptomic data and the Western blot data, especially for RING1B protein. The results showed that we have more to understand about how endometrium carries out its functions as well we have lot to discover about protein and mRNA stability inside living cells. Another surprising aspects was the lack of data regarding the number of patients diagnosed with endometriosis in India, we only have estimate but no empirical data.
Do you think these findings could eventually help in improving diagnosis, treatment, or awareness of endometriosis?
I get asked this question often, and my aim when I started work was to find out whether Polycomb group (PcG) proteins regulate EMT in endometrium and whether this regulation fails in endometriotic tissue. Now, that we have shown some correlation, we want to confirm if EMT genes are truly regulated by the Polycomb group (PcG) proteins. If we can show that, then the findings may have implications in treatment of endometriosis, since there are several FDA approved small molecules that can inhibit Polycomb group (PcG) proteins and these could be repurposed for endometriosis treatment.
What are the next steps, and what still needs to be understood before we fully uncover the molecular causes of endometriosis?
We plan to use chromatin immunoprecipitation qPCR or Chromatin immunoprecipitation sequence to assess if the Polycomb group (PcG) proteins such as RING1B, EZH1 and BMI1 can bind to promoter of key EMT and endometrial genes to regulate their gene expression. We plan to also deplete the endometrial cells of Polycomb group (PcG) proteins to assess if it affect the proliferation of these cells. By doing these experiments we would be able to add more to the literature on the molecular mechanisms involved in endometriosis.
Funding
This work was supported by the Department of Biotechnology (DBT), Govt of India (BT/PR43135/MED/97/609/2021).












