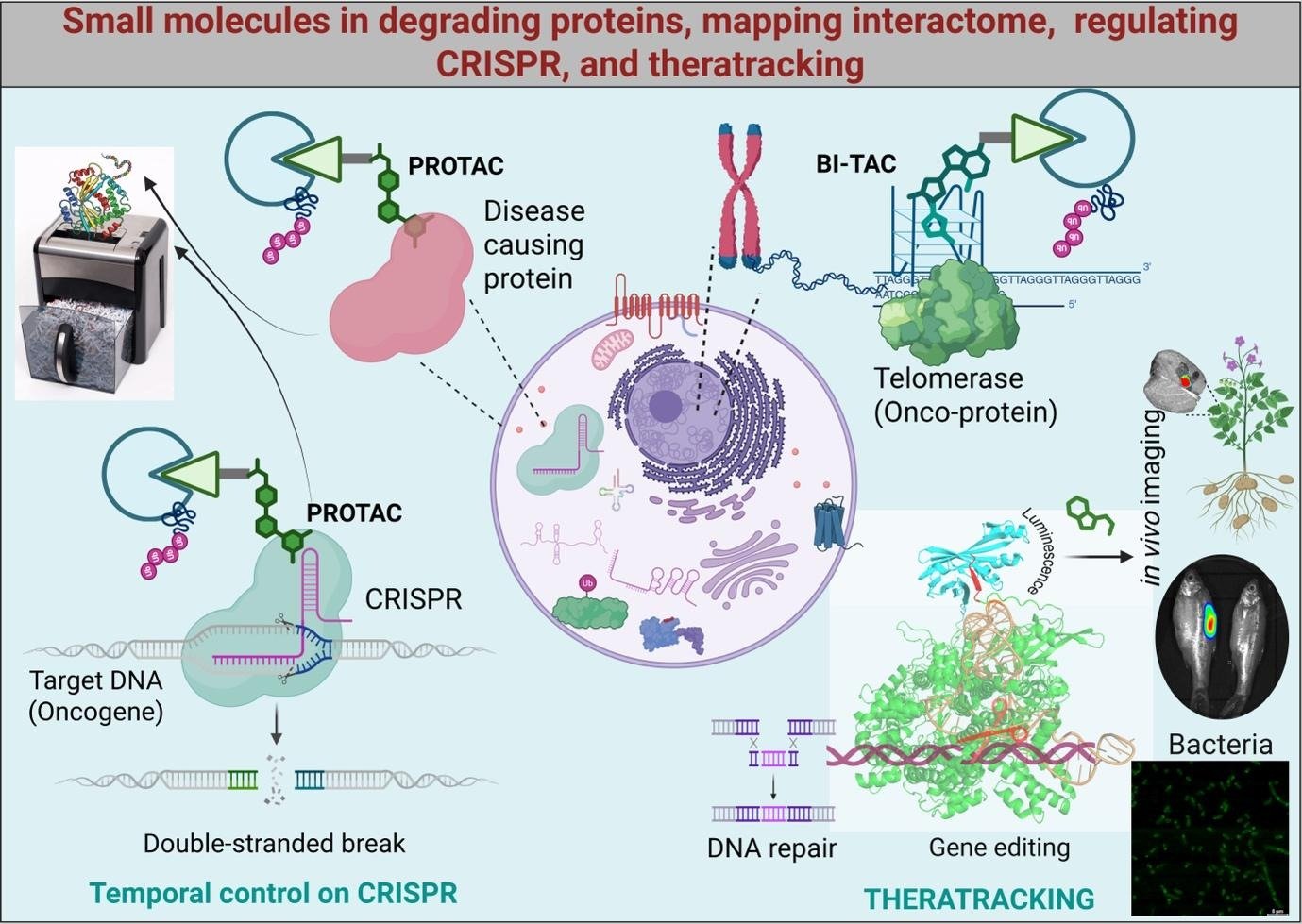
Humans are nothing but spatiotemporally regulated bags of molecules. Some are small molecules, some are large. These trillions of tiny bags can survive, often proliferate, and communicate using such different molecules. The spatiotemporal regulation of molecules inside the bags is the key to our survival and evolution. The regulation is, though, primarily controlled by the inheritable instructions stored in the form of genetic material. Mobility, motor function, and emotion are the outcomes of molecular interplay at the cellular and intercellular levels, controlled by genetics and its dynamic expression epigenetics. While nature has evolved cells into near-perfect machines for various functions, including self-replication, the embedded infinitesimal imperfection plays a key role in the evolutionary trajectory. The tiny change in genomic composition, whether through vertical or horizontal gene transfer, creates a new molecular game that often becomes a gamble for the cells. The change can benefit cells or even cause a disaster at both the cellular and the organismal levels. Genetic diseases are examples of such genetic misadventures that occur naturally during evolution. Although molecular dysregulation, compounded by environmental influences, drives human disease and suffering, it has also inspired the development of sophisticated molecular interventions aimed at restoring biological function, even sometimes enhancing performance. Molecular interventions include small-molecule drugs, nucleic acids, antibodies, and antibody-drug conjugates. Small molecules remain the majority (~80–90%) of molecular therapeutics due to their ease of development, low cost, stability, and cell permeability. Small molecules can perform a range of functions, including binding and/or inhibiting biomolecules, activating biomolecules, stabilizing or degrading biomolecules, and compartmentalizing biomolecules to achieve therapeutic outcomes. Our lab focuses on developing small-molecule agents that can engage specific proteins of interest and degrade them using the cell’s own shredding machinery, the proteasome, popularly known as PROTACs (Proteolysis-Targeting Chimeras).
We target proteins known to transform normal cells into a disease state, including cancer. Our developed bifunctional molecules can drag the harmful protein to the proteasome and enforce degradation. By degrading disease-causing proteins, one can rescue cell function and develop a therapeutic modality. Our group leveraged the potential of the PROTAC strategy and diversified the toolkit to broaden its application. For example, instead of directly engaging the target proteins, we developed molecules that can target non-protein biomolecules and degrade the interactomes of their interacting proteins. In this way, we developed a PROTAC molecule that can engage telomeric G-quadruplex DNA and target telomere-maintaining proteins, such as telomerase, which are known to be involved in tumorigenesis. Such a method enhances efficacy by multiplexing small-molecule functions, such as G-quadruplex DNA stabilization and telomerase inhibition, with telomerase protein degradation, leading to genomic instability and damage, followed by eventual programmed cell death. Our lab is developing various other PROTACs, which we call Biomolecular Interactome Targeting Chimera (BI-TAC), for targeting different biomolecules and their protein interactomes. Such PROTACs are rapidly advancing into clinical trials due to their specificity, catalytic activity, and ability to target undruggable proteins. However, certain diseases, especially hereditary genetic diseases, require more advanced therapeutic intervention for a permanent cure. Gene therapy represents a transformative approach for correcting pathogenic mutations and treating a wide range of rare genetic diseases. Some of the deadly genetic diseases include muscular dystrophies, Sickle Cell Anemia, Cystic Fibrosis, and familial hypercholesterolemia, where gene therapy appears to have the potential to achieve durable or curative outcomes. CRISPR-based genome engineering is the most advanced and effective technique for correcting defective genes. Numerous CRISPR-based gene therapy methods have been developed to address a number of genetic diseases, and many of them are in clinical trials. CRISPR has also been employed in controversial germline editing experiments aimed at introducing mutations in human embryos to confer permanent resistance to HIV; however, it raised significant ethical and regulatory concerns. Apart from unethical germline editing, off-target-mediated genotoxicity remained a serious concern in gene therapy. Spatiotemporal regulation of CRISPR activity provides an additional layer of safety, enabling more precise, controllable, and context-dependent gene therapy applications. We employ a PROTAC strategy to achieve temporally regulated CRISPR systems with improved specificity. A different kind of bifunctional small molecule has been developed that specifically binds to the CRISPR enzyme Cas9 and degrades it, turning it off. Temporal control of CRISPR activity mitigates off-target effects, thereby enhancing the precision and safety of CRISPR-based genome engineering. While one can improve target gene specificity by controlling off-target activity, tissue-specific activity is a major concern in gene therapy. In most cases, the target gene sequence is ubiquitously present across all cells in our body. Thus, a gene therapy trying to activate the tumor suppressor protein p53 in cancer tissue may also target other healthy organs and cause serious side effects. Given this, tissue-specific gene editing is crucial for achieving the intended genomic alteration or cure without causing on-target toxicity in off-target tissues or organs. While theranostics is a well-established strategy for the simultaneous diagnosis and treatment of diseases, we introduce a “theratracking” strategy for real-time monitoring of gene therapy. We developed methods for detecting the CRISPR-associated enzyme Cas9 using either a fluorogenic small molecule or a combination of protein engineering and a small molecule. For example, we developed a fluorogenic small-molecule RhoNiN that selectively binds the Cas9 enzyme via a short peptide—6X-His tag. Much like the NiNTA-based His-tag protein purification method, the fluorogenic small molecule can bind to His-tagged Cas9 to detect and probe Cas9 in real time. In a more advanced strategy, we developed an engineered thermostable Cas9 system GlowCas9 that emits bioluminescent light in the presence of a small-molecule substrate while performing gene editing. The CRISPR-associated enzyme Cas9 was genetically engineered to incorporate sticky ends derived from the deep-sea shrimp bioluminescent enzyme Nanoluciferase. While the nanoluciferase-derived sticky ends help Cas9 fold more efficiently and make it thermostable, they also generate luminescent light when in contact with a small-molecule substrate. GlowCas9 obviates the need for costly antibody-based detection of Cas9 across diverse experimental formats, including both in vitro and in vivo systems. In addition to its tracking ability, the CRISPR reporter system can efficiently disrupt target genes or repair malfunctioning genes to restore proper function in human cells. This approach enables real-time tracking of CRISPR activity and its spatial distribution within an organism, allowing timely intervention to enhance on-target specificity and improve the safety of gene therapy. Among interventions, chemical inhibitors that can block CRISPR activity and safeguard genetic materials from unintended alteration or disruption are of high priority. Our future efforts will be directed toward harnessing microbes as microreactors to develop such chemical inhibitors, thereby establishing a high-throughput, economically viable drug discovery platform. Overall, while nature explores the future course of evolution through genetic and epigenetic adventures, often entering a detrimental “Death Valley” of genetics, our lab is developing chemical interventions to rescue from such evolutionary trajectory missteps. Our lab envisions small-molecule chemical probes not as external players but as the “missing link in the central dogma”.