Why is cancer so hard to treat? This is a question that patients, families, and even doctors ask every single day. One of the most painful truths about cancer is that even after successful treatment after surgery, chemotherapy, or radiation the disease sometimes comes back. This recurrence or relapse is often more aggressive and far more difficult to treat than the original tumor.
But what exactly causes this recurrence? Why do some cancer cells manage to escape treatment and return months or even years later? These questions drive the core of the research in our laboratory, and today I want to take you through what we have discovered.
The Hidden Enemy: Dormant Cancer Cells: After treatment, most cancer cells die. But a small group often just a handful manage to survive. They may hide in our body for years in a dormant or “sleeping” state. During this time, they don’t divide, they don’t spread, and they don’t trigger symptoms. They remain silent.
But eventually, something awakens them.
Scientifically, this awakening is referred to as the acquisition of self-renewal ability. In simpler terms, these dormant cells regain the ability to behave like stem cells cells that can divide endlessly and create new cancer cells. These cancer cells are generally called Cancer stem cells (CSCs). Once this switch turns on, recurrence becomes likely. Our lab focuses on what causes this switch. How do sleeping cancer cells reprogram themselves to start dividing again? What signals do they receive? And more importantly is it possible to block this process?The External Influence: How the Microenvironment influence cell behavior: One possibility is that the awakening comes from within the cancer cell itself its inherent properties. Another possibility is that it is triggered by signals from the environment where the cell is hiding. Though both mechanisms can contribute, now we will talk about the signals from the surroundings. Dormant cancer cells are influenced by their microenvironment, the surrounding cells, proteins, and chemical signals in the tissue where they hide. Normal cells constantly communicate with their neighbors through signaling pathways. One of these pathways is the Eph/Ephrin pathway, which involves interactions between a receptor (Eph) and its ligand (Ephrin).
Usually, this pathway mediates communication between two neighboring cells. When Eph and Ephrin interact across two different cells, signals are triggered in both. When neighboring cells interact, the ligand sends signal to the receptor and the receptor initiates a signaling cascade to bring about a function. This signaling downstream of the receptor is called “forward signaling”. This is especially important in places like stem cell niches, where one cell remains dormant while its neighbor begins to differentiate.
For example, EphrinB1 interacting with EphB1/2 helps regulate the maintenance and differentiation of intestinal stem cells.
Cancer is an Enigma: Apart from the regulation of normal stem cells, Eph/Ephrin pathway plays a role in cancer as well. When neighboring cells interact, the ligand sends signal to the receptor to initiate “forward signaling”. Though this ligand-induced “forward signaling” suppresses cancer progression (by blocking proliferation and cell spread), the Eph receptors adopt alternate signaling mechanism to favor cancer progression- by interacting with other pathway receptors. Through this altered pathway or “noncanonical signaling”, the receptors (Ephs) favor the progression of cancer by augmenting proliferation, vascularization and expanding cancer stem cell (CSC) pool.
The Ephrins on the other hand are considered to be suppressing cancer, as they initiate tumor suppressive “forward signaling”. When we do experiments in lab, in a petri dish, we see that ligands inhibit cell proliferation and spread of the cancer cells. So, it should be limiting the growth of the cancer and spread of the disease. But, when we analyzed the role of this pathway in cancer, patients who had high expression of the ligand showed poor outcome, implying that the ligands are doing some other functions, favoring cancer progression.
What do the ligands do to worsen the cancer? This was a question that was not answered for a long time.
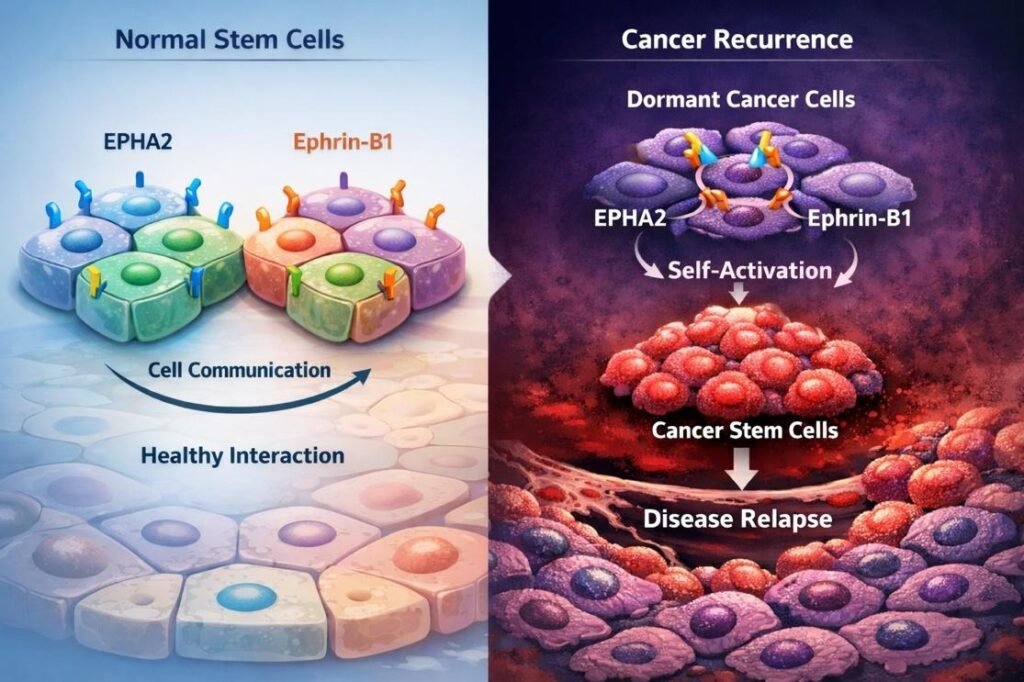
A Surprising Discovery: The Cancer Cell Talks to Itself: Our lab found something unexpected and intriguing.In the cancer cells that acquire self-renewal ability, the Eph/Ephrin interaction does not happen between two different cells. Instead, it occurs within the same cell.
This is unusual and not seen in normal cells or even in other types of cancer cells. Even more fascinating, the combination of receptor and ligand involved in this self-interaction is unique. We discovered that Ephrin-B1 interacts with EPHA2 within the same cancer cell to trigger self-renewal. This autocrine or “self-activating” signaling mechanism may be one of the key drivers of recurrence.
Origin of the Story: In our effort to understand how “sleeping” cancer cells wake up and become more aggressive, we studied the signals that control this process. These awakened cells can turn into cancer stem cells (CSCs) a small but powerful group of cells that can drive tumor growth and recurrence.To investigate this, we used advanced techniques like mass spectrometry to analyze proteins on the surface of cancer cells and the internal signaling pathways they use to communicate. Through this, we identified an important pathway called the Eph/Ephrin pathway, and in particular, a molecule called Ephrin-B1.Our early experiments with oral cancer cells suggested that two proteins EPHA2 (a receptor) and Ephrin-B1 (a ligand) play a key role in this process.
An Unusual Partnership: In many cancer cells, Ephrin-B1 is usually not present on the cell surface and hence not engaged in signaling. Meanwhile, EPHA2 is highly active through the “noncanonical signaling” promoting tumor growth, including proliferation and formation of new blood vessels. However, when the surrounding environment sends signals that encourage cancer cells to behave like stem cells, Ephrin-B1 moves to the cell surface. This allows it to interact with EPHA2.What makes this surprising is that these two proteins don’t normally pair up. They belong to different subgroups, and typically, ligands and receptors interact within their own group. This unexpected pairing caught our attention.
Proving the Interaction: To confirm that EPHA2 and Ephrin-B1 truly interact, we used several laboratory techniques (Co-immunoprecipitation, Proximity Ligation Assay, Foster Resonance Energy Transfer etc.) that allow us to detect close protein interactions inside cells. These experiments showed that the two proteins do indeed bind to each other and importantly, this happens within the same cell.We also found that this interaction plays a major role in increasing the number of cancer stem cells. We confirmed this using multiple experimental approaches, including studies in cell cultures, animal models, and samples from oral cancer patients.
“Self-Talk” That Reactivates Cancer Cells: When cancer cells receive signals to “wake up,” Ephrin-B1 moves to the cell surface and interacts with EPHA2 on the same cell. This creates a kind of self-communication, where the cell activates its own signaling system.In this case, the signal flows in an unusual direction from the receptor to the ligand triggering what scientists call “reverse signaling.” them to CSCs. Interestingly: The usual (“forward”) signaling helps slow down tumor growth. But this reverse signaling has the opposite effect it promotes the awakening of dormant cancer cells and helps turn them into cancer stem cells.
Why This Matters: Our findings reveal a new way that cancer cells can activate themselves and become more aggressive. While this study focused on oral cancer, similar mechanisms may exist in other types of cancer and even in normal stem cell systems.From a treatment perspective, this discovery is important because cancer stem cells are often responsible for tumor relapse and resistance to therapy.We found that the unusual interaction between EPHA2 and Ephrin-B1 may disrupt normal cellular balance and contribute to these problems. This opens up the possibility of developing new treatments that specifically block this interaction.Looking Ahead: We are now working on designing drugs that can prevent this unusual protein interaction. By blocking this “self-talk,” we hope to stop dormant cancer cells from waking up and becoming dangerous again.With further research, this approach could lead to new strategies to prevent cancer recurrence and improve long-term treatment outcomes.