What inspired you to combine probiotics with nanoparticles, and how did this research idea begin?
For thousands of years, Indian households have used curd, buttermilk, and fermented foods not just for taste, but for health. Our ancestors knew that friendly bacteria (probiotics) keep the gut healthy and the body strong. My own research began with a simple question: Can we make these friendly bacteria even more powerful using modern science?
I have spent over a decade studying Lactobacillus strains the same family of bacteria found in yogurt. I discovered two new strains from sheep milk, deposited them in India’s National Centre for Microbial Resource, and even helped develop a probiotic biscuit that reached the Indian market. But I wanted to go further.
At the same time, scientists around the world were exploring nanoparticles particles so small that thousands could fit on a single hair. Iron-based nanoparticles are known to kill harmful bacteria, but they can also be toxic to our own cells.
Then came the “eureka” moment: What if we coat the probiotic with nanoparticles? The probiotic would act as a living, protective base. The nanoparticles would add extra killing power. Together, they might be stronger, safer, and smarter than either alone.
That is how the nanobiohybrid was born not in a distant Western lab, but right here in Kolhapur, India, combining ancient wisdom with 21st-century nanotechnology.
Can you explain in simple terms what a nanobiohybrid is and how it works?
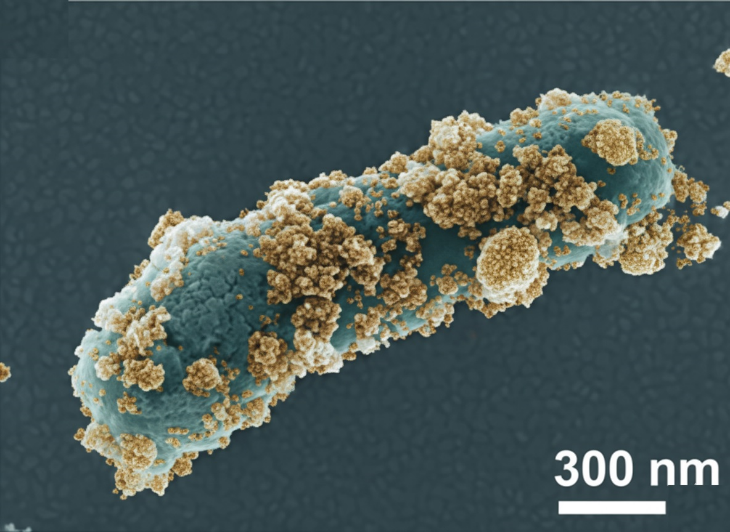
Imagine a friendly soldier a probiotic bacterium that already knows how to fight bad germs. Now imagine giving that soldier a special armor made of ultra-tiny magnetic particles (maghemite, a form of iron oxide). That armored soldier is a nanobiohybrid. Here is how it works in simple steps: Component Role Probiotic (Lactobacillus rhamnosus) Lowers pH, secretes natural antibacterial chemicals, and is already safe for humans. write inparagraphs withut chsniung many words Maghemite nanoparticles (γ-Fe₂O₃) Generate reactive oxygen species think of them as “oxidation bombs” that destroy bacteria and break down sticky biofilms Nanobiohybrid (both together) The probiotic guides the nanoparticles; the nanoparticles boost the probiotic. Together, they kill more bacteria, break more biofilms, and scavenge harmful free radicals The two components are held together by electrostatic forces like opposite sides of a magnet. We simply mix them in water, and they self-assemble. No harsh chemicals. No complex machinery. Simple, elegant, and effective.
Your study shows improved antibacterial and antibiofilm activity. Why is this important for real-world problems like infections and wound healing?
Most people think bacteria float around freely. In reality, most harmful bacteria stick to surfaces medical implants, catheters, wounds, teeth, even kitchen counters and form a slimy, protective layer called a biofilm. You can think of a biofilm as a fortress. Inside that fortress, bacteria can become up to 1,000 times more resistant to antibiotics. This explains why chronic wounds often do not heal, why urinary catheters can lead to repeated infections, and why some infections seem extremely difficult to treat. Our nanobiohybrid tackles this problem in two important ways. First, it prevents biofilm formation, stopping bacteria from building the “fortress” in the first place. Second, it can eradicate existing biofilms, breaking down the structure even after it has already formed. In our tests, the nanobiohybrid was especially effective against Listeria monocytogenes, a dangerous foodborne pathogen, and Staphylococcus succinus, a known biofilm-forming bacterium. At the highest concentration, it reduced biofilm formation by nearly 80%. This has important real-world implications. Chronic wounds, such as diabetic ulcers, often fail to heal because of persistent biofilms; a spray or gel containing this nanobiohybrid could improve treatment outcomes. Medical devices like catheters, stents, and implants could be coated with it to prevent infections without relying heavily on antibiotics. In food safety, the nanobiohybrid could be incorporated into packaging to limit pathogen growth. Finally, because it works through multiple mechanisms rather than a single pathway, bacteria are less likely to develop resistance, making it a promising strategy in the fight against antibiotic resistance.
Fig. Image of Probiotics and Probiotics coated with Nano-particle (graphically generated image to know the concept in simple way)
What was the most surprising or exciting result you observed during your research?
Three results genuinely surprised and excited us.
First, the nanobiohybrid was much safer than nanoparticles alone. When we tested the nanoparticles by themselves on human intestinal (Caco-2) cells, cell viability dropped to about 62% at the highest concentration. However, when combined with the probiotic to form the nanobiohybrid, cell viability remained above 86%. The probiotic essentially acted like a protective cloak, shielding human cells from the harsher effects of the nanoparticles. Second, the probiotic did not die in fact, it became stronger. We initially expected that coating the bacteria might reduce their survival. Instead, Lactobacillus remained fully alive. Even more interesting, the coated probiotic showed improved tolerance to stomach acid and bile, suggesting it could survive longer if administered orally.Third, the hybrid was able to break down mature biofilms, not just prevent them. Many studies focus only on preventing biofilm formation, which is like checking whether a lock can stop a thief from entering. But what happens if the thief is already inside? We addressed both scenarios. The nanobiohybrid not only prevented biofilms from forming but also disrupted already established ones reducing them by nearly 70% in some cases.
These results told us: This is not just another lab curiosity. This could become a real product.
Do you think this nanobiohybrid approach could have future applications in medicine, healthcare, or industry?
Absolutely. I see at least five major application areas for this technology.
One important area is healthcare, especially in treating chronic wounds such as diabetic ulcers. Because biofilms often prevent healing, a nanobiohybrid-based spray or gel could help clear infections and promote faster recovery.
Another key application is in medical devices. Catheters, stents, and implants are highly prone to biofilm-related infections. Coating these surfaces with the nanobiohybrid could significantly reduce infection risk without relying heavily on antibiotics. Food safety is also a promising field. The nanobiohybrid could be incorporated into food packaging materials to prevent the growth of harmful pathogens like Listeria, helping to extend shelf life and improve safety. A fourth area is gut health and probiotic therapy. Since the coated probiotic shows improved tolerance to stomach acid and bile, it could be developed into advanced probiotic formulations that survive longer and function more effectively inside the body.
Finally, this approach could contribute to combating antibiotic resistance. Because the nanobiohybrid works through multiple mechanisms both biological and chemical it becomes much harder for bacteria to develop resistance compared to traditional antibiotics.A word of realism is important. Before any of these applications become real products, we need to complete animal studies followed by human clinical trials. Regulatory pathways for nanobiohybrids are still not clearly defined, as they do not fit neatly into existing categories like drugs, medical devices, or probiotics. However, this also presents an opportunitycountries like India could take the lead in shaping new regulatory frameworks for such innovative hybrid technologies.
What are the next steps in your research, and what challenges still need to be addressed?
Our immediate next step is to move from the lab bench to living systems. So far, we have completed test-tube (in vitro) and cell-culture studies. The next phase involves animal studies to understand whether the nanobiohybrid works effectively inside a living body, how long it remains active, and whether it causes any hidden toxicity over time. We are currently designing experiments using small animal models, particularly mice, focusing on applications in wound healing and gut health. At the same time, several important challenges need to be addressed. One of the biggest is scale-up. While producing small quantities in the lab is manageable, scaling up to industrial levels while keeping the bacteria alive and ensuring uniform attachment of nanoparticles is much more complex. Stability is another concern, as this is a living material that must remain viable during storage, transport, and handling. Regulatory pathways also remain unclear nanobiohybrids do not fit neatly into existing categories such as drugs, medical devices, probiotics, or nanomaterials, both in India and globally. Cost is equally critical; although nanoparticle synthesis is becoming more affordable, the final product must be accessible to ordinary people, not just high-end healthcare settings. Despite these challenges, I remain optimistic. India has a strong heritage of probiotic use, a rapidly growing biotechnology sector, and a government that actively supports innovation and startups. Having received recognition such as the Start-Up Hero of Maharashtra award under the Make in India initiative, I believe that with the right collaborations and partnerships, this research can be translated into a practical product that benefits millions of people.