The rotation of the Earth around its own axis causes alternating periods of light and darkness and the 24-hour duration of this day-night cycle has remained approximately constant over several hundred million years. Life on Earth has learned to adapt to this day-night cycle through an internal clock called the circadian rhythm (from Latin: circa meaning around, and diem, a day) (Figure 1A). The circadian clock provides organisms with an evolutionary advantage by allowing them to anticipate the onset of light and darkness rather than merely respond to changes in illumination. We most commonly perceive the existence of this clock in our day-to-day life through jetlag, which occurs when we travel across time zones and throw the clock out of synchrony. An important property of the circadian clock is that it can be entrained by external cues such as light and temperature (known as Zeitgebers, German for time-givers), which is why jetlag disappears once our body has accustomed itself to the new time zone. The discovery of the molecular gears that keep the clock ticking was honoured by the 2017 Nobel Prize in Physiology or Medicine awarded to Jeffrey C. Hall, Michael Rosbash, and Michael W. Young. Somewhat alarmingly, disruption of the circadian clock because of ever-increasing shift work, as well as widespread exposure to low levels of light at night, has been linked to a range of maladies such as cardiovascular disease and cancer. There is thus an urgent need to understand the clock using both bottom-up and top-down research strategies.
“Our body’s internal clock runs on a delicate balance of molecular interactions, where tiny changes in protein behavior can shape the rhythm of life.”
Our laboratory at the Molecular Biophysics Unit in IISc Bangalore studies biological systems where the jiggling and wiggling of molecules (referred to as dynamics), and not merely their shape (their structure), is important for carrying out function. Since the inception of the lab in November 2017, we have been keenly interested in the circadian clock, because the spatial and temporal choreography of the various protein molecules constituting the circadian clock is vital for maintaining its approximately 24-hour periodicity. The core circadian oscillator is made up of four proteins, PER2, BMAL1, CRY1 and CLOCK. We chose to initially focus on PER2, because it is partially structured and partially disordered, and its lifetime inside the cell is critical in setting the 24-hour time period of the circadian clock. The lifetime of PER2 is, in turn, controlled by the presence or absence of phosphoryl groups on serine residues located at the boundary of the ordered and disordered regions. Once the phosphoryl group gets attached (through an enzyme-catalysed process called phosphorylation), it becomes easier to degrade PER2, although why this must be so is not clear.
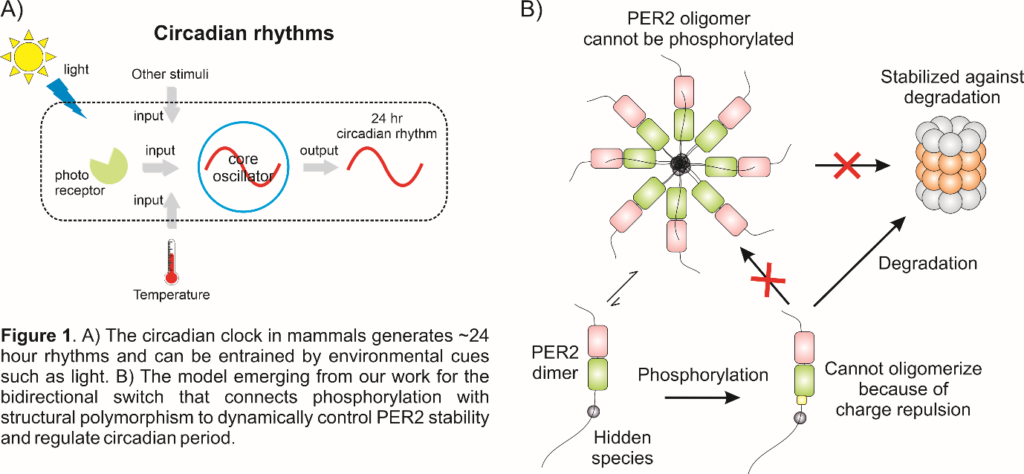
In 2019, Ms. Diksha Sharma, a project assistant in the lab, made the serendipitous discovery that PER2 does not exist in isolation, but instead forms large assemblies (called oligomers), each containing ~30 PER2 molecules, that could be visualized under an electron microscope. This observation led us to formulate an exciting hypothesis – that the serines which had to be phosphorylated are buried inside the oligomer. Once the serines get phosphorylated, the negative charges on the phosphoryl group are thrown in close proximity to each other and repel each other to break the oligomer apart. The smaller pieces would then degraded by the cellular machinery. Although this hypothesis gave us a way to explain why it was easier for the cell to degrade phosphorylated PER2, it raised as many questions as it answered – if PER2 oligomers protected the sites of phosphorylation, how would PER2 get phosphorylated in the first place? Would a single phosphorylation be enough, or would multiple serines have to get phosphorylated before the oligomer disintegrated?
In 2020, Ms Paulomi Sanyal, a talented doctoral student, took over this challenging project and it required all her tenacity and perseverance to bring it to fruition. Our laboratory has traditionally used nuclear magnetic resonance spectroscopy (NMR, a method closely related to magnetic resonance imaging) to characterize the dynamics of biomolecules and study their impact on function. However, Paulomi recognized the value of integrating information from several biophysical methods such as light scattering, analytical size-exclusion chromatography, circular dichroism, fluorescence, thermodynamic analysis, NMR spectroscopy and electron microscopy to tackle the complexity that accompanied the questions we had posed.
The outcome of this effort is a unified molecular model for how phosphorylation of PER2 modulates circadian timekeeping (Figure 1B). PER2 forms large oligomers and the stability of the oligomer derives from cooperativity between the structured and the disordered regions. The region that is phosphorylated is protected within the oligomer and cannot be phosphorylated, with the result that PER2 is stabilized and its lifetime in the cell increases. Crucially, there is also a small hidden population of PER2 that is dimeric (i.e. each dimer contains only two molecules of PER2), and PER2 molecules can efficiently jump back and forth between dimeric and oligomeric forms. It is this dimer that is phosphorylated and eventually targeted for degradation. Assemblies formed from phosphorylated PER2 are much less stable and systematically reduce in size as the amount of negative charge per PER2 increases. The consequence is a two-way bidirectional switch where oligomerization prevents phosphorylation and the addition of phosphate groups destabilizes the oligomer. The balance between these two opposing driving forces eventually controls the cellular lifetime of PER2 and thereby influences the circadian time period. It is noteworthy that this switch hinges on an ‘invisible’ dimeric state of PER2, highlighting the importance of characterizing these latent states in order to understand function in its entirety.
“From hidden protein states to dynamic molecular switches, the circadian clock reveals how life keeps time at the smallest scale.”
One of the quintessential goals of biophysics is to explain biological function from a molecular perspective. The conclusions which emerge from our work accomplish this by explaining why phosphorylation ‘destabilizes’ PER2 and pushing the boundaries of our understanding of the molecular mechanism controlling the circadian clock. The results link structural polymorphism in PER2 to post-translational modifications (a broad term that encompasses chemical changes to a protein after its synthesis or translation) and identify a bidirectional molecular switch that dynamically regulates the circadian period. Exciting recent work from other labs across the world have also demonstrated the existence of large assemblies containing PER2 inside living cells, bridging our ‘test tube’ science with the behaviour of living organisms. During this work, we had the pleasure of working alongside Prof. Carrie Partch, a world leader in the biophysics of the circadian clock, and her team from the University of California Santa Cruz. Ms. Diksha Sharma, who began her adventures into the circadian clock with us, is currently a doctoral student in the Partch lab. We are also grateful to the generous and flexible funding provided by the DBT/Wellcome Trust India Alliance for this work and to Prof. Jayant Udgaonkar from IISER Pune for encouraging us to persevere with this project.