The inspiration
In 2006, Sammual I. Stupp delivered series of lectures titled “Self-assembled peptide amphiphiles nanofibers with extensive applications” at University of Bordeaux, France. It gave me adventures and pleasant introduction into the world of supramolecular gel for diverse applications.
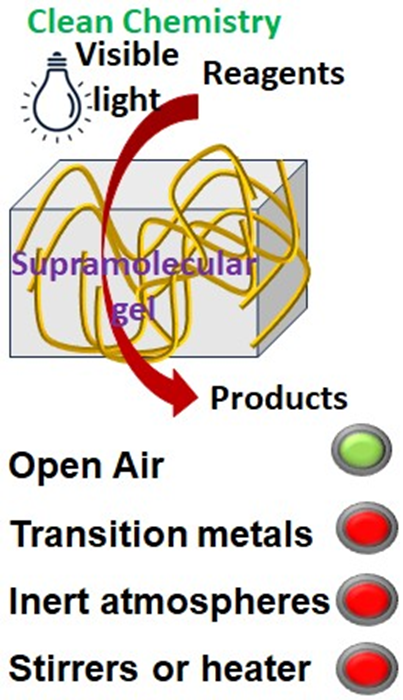
My research at Department of Chemical Sciences, Indian Institute of Science Education and Research (IISER) Kolkata focusses on development of designer amino acids analogues and peptide mimetics as supramolecular gelator for reaction engineering and visible-light organophotocatalysis with low-cost starting materials and open air. We design and synthesize chemically inert peptide mimetics that are capable of forming self-assembled gels in diverse solvents in the presence of various reactants as well as reagents. Our goal is to develop synthetic gelator that have no chromophore system and will pass the light to the photocatalysts. The instant gel should be formed by simple heating-cooling cycles and sonication, as evident by the lack of gravitational flow when the vial was inverted at ambient condition. The gel should be robust, stable for couple of days, transparent or translucent and the gel-to-sol transition temperature (Tgel) should be comparatively high. Additionally, the gel should dynamic and have significant self-healing properties i.e. when the existing structures of the gel are disrupted, new self-assembling structures spontaneously evolved. The peptide mimetic gel impedes oxygen diffusion inside the gel, protect the reagents form decays and allow air-sensitive reaction under open air conditions.
Why gel as reaction medium?
Day to day, our award-winning chemical reactions are becoming more complicated, needs expensive instruments support such as Senk line, chemical hoods, rotary evaporator, recirculating chiller, low temperature bath, glove box, high vacuum pomp, microwave reactor, sonication bath, dehumidifier, pH meter, specialized glass setup, inert atmosphere, high temperature, long time reflux. Moreover, the starting materials, catalysts, co-catalysts, initiators, dry solvents are also very expensive and beyond the reach of our school and undergraduate degree college laboratory. In this situation, it is very difficult to demonstrate those modern experiments for the students and students are forced to memorising the name reactions and chemical equations and being demotivated to learn chemistry. To overcome this, I look into nature. Nature is doing all kind of selective and specific chemical reactions inside soft tissue with extreme precession. Following that, I have developed soft material such as gels that can behave like a matrix and accommodate various reactants as well as reagents, impedes oxygen diffusion inside the system to protect the transition states and intermediates and allow air as well as moisture sensitive reaction under ambient conditions. Moreover, the gelators can be recycle and reused for sustainable future.
My vision
Eventually, we utilize a supramolecular gel matrix to confine the reactants and block oxygen diffusion, allowing air-sensitive catalytic EnT-mediated dearomative [4+2] cycloaddition of naphthalenes with styrenes under aerobic conditions. Also, we have performed a redox-neutral trifluoromethylation of dienes using g-C3N4 as a visible-light organophotocatalyst inside a supramolecular gel matrix in open air. The field is wide, interesting, challenging and filled with high prospect and now is the best time to explore.