What first led you to investigate this unusual presentation of asterixis in your patient?
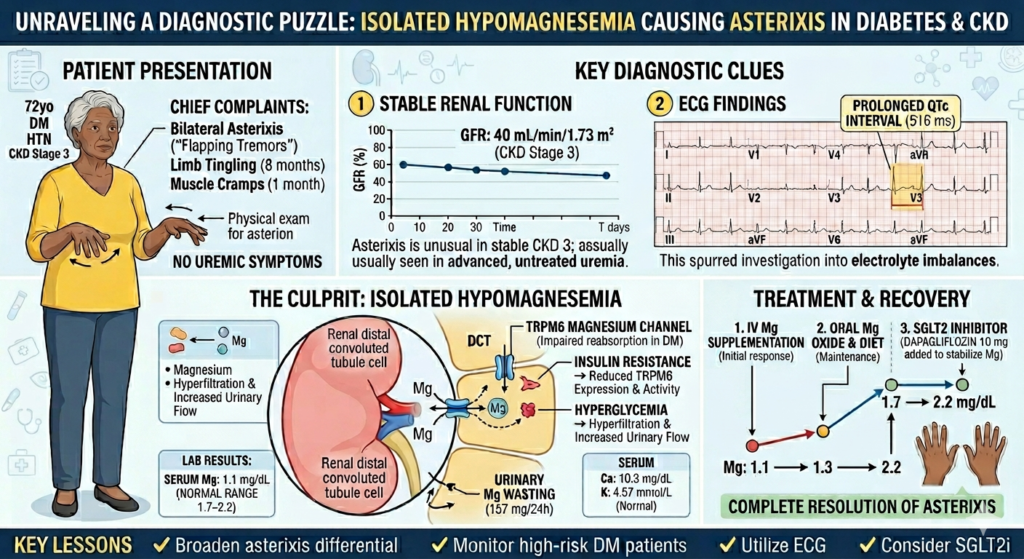
The investigation into this patient’s unusual presentation of asterixis was driven by several clinical inconsistencies. In a patient with chronic kidney disease (CKD) generally presence of asterixis suggests uremic encephalopathy and requires haemodialysis. But in this case patients eGFR and serum creatinine levels were stable; urine output was maintained and there were no symptoms and evidence of uraemia.
Also, the patient, an elderly woman with type 2 diabetes and hypertension, presented with neurological symptoms including tingling sensation in all four limbs (for 8 months) and muscle cramps (for 1 month). Yet upon examination her neurological findings were normal.
Her electrocardiogram (ECG) showed a significantly prolonged QTc interval of 516 milliseconds which prompted further evaluation.
Can you explain in simple terms what asterixis is and why it is usually linked to more severe conditions?
In simple terms, asterixis is a movement disorder characterized by an inability to maintain a fixed posture due to involuntary interruption in muscle contraction. It is often referred to as a “flapping tremor” because it looks like a bird flapping its wings when a patient tries to hold their hands out with the arms extended and wrists dorsiflexed. Technically, it is not a true tremor and it is a form of negative myoclonus. This means that instead of an active shaking, there is a brief, sudden loss of muscle tone that causes the hand to drop momentarily before the brain quickly corrects the position.
Asterixis typically signifies a significant disruption in the brain’s ability to regulate motor control and are therefore are linked to more severe conditions that interfere with the central nervous system’s signalling like –
-Organ failure associated metabolic encephalopathies including liver failure, kidney failure and respiratory failure
-Structural brain damage to specific areas of the brain, such as the thalamus or midbrain, which are responsible for coordinating movement and posture.
-Severe Electrolyte imbalances such as hypomagnesemia identified in this case, can disrupt the electrical stability required for normal muscle tone and posture
What made this case different, and how did you identify hypomagnesemia as the underlying cause?
The patient an elderly woman with type 2 diabetes, diabetic nephropathy and hypertension had neurological manifestations and bilateral asterixis. What made this case different was a stable renal profile without uraemia and normal neurological examination and imaging.
Her liver function test and standard serum electrolyte panel was normal and no history of drugs causing asterixis was found.
Her ECG showed a prolonged QTc interval of 516 milliseconds, prompting evaluation of serum calcium and magnesium levels. The results revealed a serum calcium of 10 mg/dL (corrected calcium was 10.32 mg/dL) and a serum magnesium of 1.1 mg/dL which was reduced.
As hypomagnesemia is induced by two major mechanisms: gastrointestinal or renal losses, she underwent further evaluation for the underlying cause. The underlying cause of the depletion was ultimately attributed to diabetes mellitus–related renal magnesium wasting, as her 24-hour urinary magnesium was significantly elevated and other renal causes of hypomagnesemia were ruled out.
The diagnosis was also confirmed when the patient’s asterixis resolved completely following magnesium supplementation.
What surprised you the most during this diagnostic process?
Magnesium deficiency usually occurs as part of a complex electrolyte imbalance involving low potassium or calcium. In this case, the patient’s potassium and calcium levels were completely normal making it a case of Isolated magnesium deficiency, which is rare.
It was striking to find that her diabetes was the primary cause of such severe magnesium wasting. Despite the body being deficient, her kidneys continued to lose magnesium at a high rate (157 mg/24 hours), highlighting how diabetes can actively disrupt mineral homeostasis.
Another rewarding surprise was the complete resolution of the flapping tremors once the magnesium was replaced, confirming that a seemingly complex neurological presentation could be fixed with a straightforward metabolic correction.
Why is magnesium deficiency often overlooked, especially in patients with diabetes?
The prevalence of hypomagnesemia is approximately 2% in the healthy population while in Type 2 diabetes, it is between 9.1% and 47.7%. Henceforth, hypomagnesemia occurs about ten times more frequently in diabetics than in the general population.
Magnesium deficiency is frequently overlooked in clinical practice, particularly in patients with diabetes, for several reasons:
Magnesium is not typically included in a standard, routine electrolyte panel, meaning it is often not measured unless a clinician specifically orders it.
Neurological symptoms of hypomagnesemia, such as paraesthesia like tingling and muscle cramps, can be assumed to be due to diabetic peripheral neuropathy.
Lack of specific symptoms or physical signs that point exclusively to hypomagnesemia makes it harder to identify.
There is currently a lack of organized consensus guidelines to help clinicians screen and manage for hypomagnesemia systematically.
What are the key lessons for clinicians and what further research is needed in this area?
This case highlights the importance of evaluating for underlying causes of asterixis, as well as the predisposition of individuals with diabetes mellitus to renal magnesium wasting. Hypomagnesemia, although frequently overlooked, is a clinically important electrolyte disturbance that can present with diverse neurological, cardiovascular, endocrinal and muscular manifestations. Identifying this condition presents a significant diagnostic challenge, yet it offers the potential for a complete clinical recovery. Consequently, early recognition and prompt correction are essential to achieving a positive outcome and preventing further complications. Further research is needed in this area as there is currently limited data and a lack of organized consensus guidelines to guide clinicians on how to systematically screen for and manage hypomagnesemia.