Bone defects caused by injuries, diseases, and congenital disorders possess significant challenge in the healthcare sector whenever the extent of damage is beyond the natural healing response and capacity of human body. The conventional treatment methods of bone injuries that require regeneration of new bone tissues include autografts and allografts often found to be failed due to lack of donors, risk of infections and the threat of immune rejection. To avoid the chances of graft failure, researchers are now focussing on developing new treatment strategies with the help of tissue engineering using advanced design and biomanufacturing techniques for making bio-compatible materials to replicate the native microenvironment of body’s natural wound healing process. The three-dimensional matrix (scaffold) for cell growth in tissue engineering can be fabricated through various methods such as electrospinning, 3D bioprinting, freeze drying etc. Scientists are utilizing such bioengineered scaffolds using advanced materials (such as polymer, ceramic, and nanomaterials) to achieve a supply line where on-demand organ design and fabrication would be a reality. The primary objective of such approaches in bone-tissue regeneration is to mimic the native extracellular matrix (ECM), which supports and guide cell growth and ensures faster healing rate. The ECM is made up of fibrous proteins, glycosaminoglycans and signalling molecules which act as a support system and helps in the cell migration, differentiation and proliferation.
Electrospinning has gained more interest in this regard due to its ability to make ultra fine porous fibre structures with diameters similar to natural collagen fibrils. These nanofibrous scaffolds have high surface area, tunable physical properties and interconnected pores ideal for supporting cell growth. However, it fails to deliver some important requirements such as mechanical strength, nanoscale pore size which limits oxygen and nutrient transport, cell infiltration and vascularization. Hence, recent studies are more focused on improving the mechanical and physico-chemical properties of the fibrous electrospun scaffolds through chemical functionalization and crosslinking to improve the cell attachment and nutrient transport. Additionally, the healing process can be accelerated by incorporating natural bioactive agents and growth factors and multiple functionalities can be gathered by designing multilayered scaffolds through electrospinning technique. This technique has actively helped to make bioactive ECM mimicking scaffolds with superior and faster healing performance.
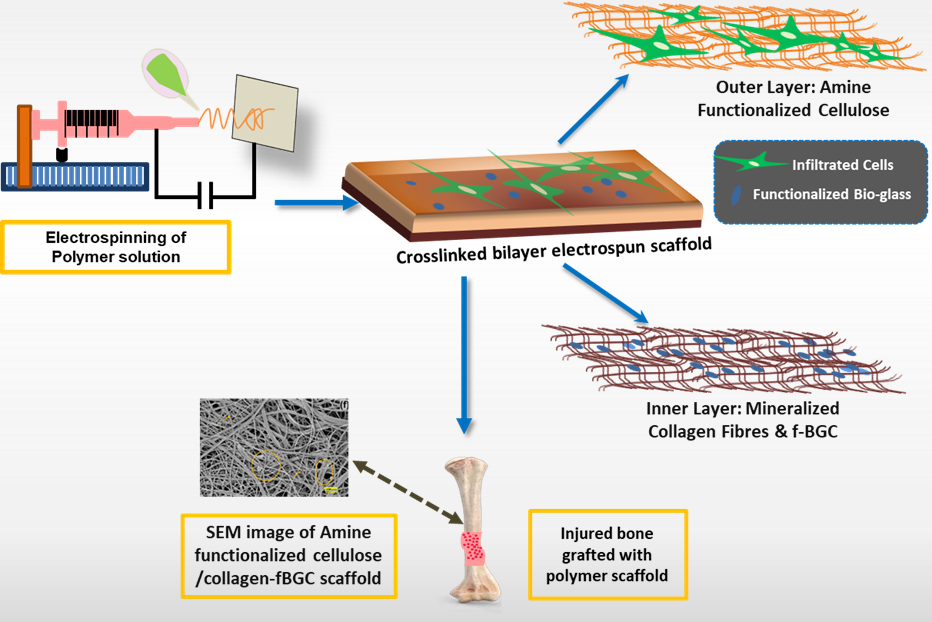
A recent study by our group at “PRINT” lab of JISIASR, JIS University in collaboration with Jadavpur University, IIEST-Shibpur, and Manipal Academy of Higher Education, demonstrated that a polymer based bilayered functionalized fibrous scaffold composed of cellulose and collagen via electrospinning shows promising result in better healing of bone injury by imparting bone tissue regeneration at the defect site. Owing to its acceptable mechanical properties and surface functionalities, electrospun cellulose fibers exhibit a good cell adhesion. In this study, the regenerated cellulose scaffold was first oxidized to modulate the degradation and improved cell attachment. Subsequently, amine functionalization was carried out through the conjugation of L-arginine onto the cellulose surface via EDC/NHS coupling reaction, enhancing the surface hydrophilicity and positive charges that facilitate protein adsorption.
The amine functionalization ensured the better expression of osteocalcin and alkaline phosphatase (ALP) indicating enhanced bone regeneration potential. To further mimic the native bone healing environment, a second layer of collagen containing functionalized bioactive glass-ceramic (f-BGC) was developed representing the organic and inorganic components of bone tissue. The f-BGC can function like hydroxyapatite, the natural inorganic component of bone and it releases bioactive ions which can stimulate osteogenesis. Furthermore, functionalization of bioactive glass particles with glutamic acid enhanced the radical scavenging ability as compared to silica and hydroxyapatite. The morphology studies revealed porous structures that facilitates cell infiltration and calcium deposition. The bilayered scaffold with optimized functionalization and porosity supports efficient nutrient transport and cell infiltration. The cytocompatibility studies showed excellent viability percentage for multiple cell lines and a 70% increase in pre-osteoblast differentiation within three days. Although these smart scaffolds have indicated highly promising results, our ongoing research is focused on fine tuning of scaffold’s mechanical properties, which are considered as critical factors for maintaining the structural stability under various physiological stress throughout the healing process.
Ongoing research in this area has been more directed toward the development of smart multifunctional scaffolds consisting of both biological compatibility and mechanical stability. Considering the dynamic environment of wound throughout the healing process, a smart scaffold responsive to multiple stimuli including pH, temperature, enzymes and certain physico-chemical cues has offered significant improvement when the fabrication process is combined with advanced fabrication techniques such as electrospinning, 3D and 4D printing. The future of biomanufacturing and scaffold fabrication lies in the digitization of patient-specific data obtained from imaging techniques such as CT and MRI. This in-turn allows simple design and fabrication of scaffolds as per required size and geometry of the injury point for better compatibility and integration with the surrounding tissue. For successful clinical implementation of such scaffold-based strategies, intensive studies under several physiologically relevant conditions are essential.
Recent initiatives by at several government and private sectors to build a strong partnership of many medical institutes and hospitals with the research institutions place India in a favourable position to bridge the gap between laboratory research and real-life applications. Over the past decade, our group has been trying to integrate multiple biomanufacturing methods to overcome the existing limitations of individual fabrication techniques aiming to develop translational technologies for bone and skin tissue regeneration as a step forward toward artificial organ design and fabrication. We extend our sincere thanks to JIS Group and our funding agencies Anusandhan National Research Foundation-ANRF (earstwhile SERB), Government of India for supporting this research.